States of Matter - GCSE Chemistry
Introduction
- Matter is anything that has mass and takes up space. Everything around us —
- In this topic, you will learn about the three states of matter (solid, liquid, and gas), how they change from one state to another, and the difference between physical and chemical changes.
- Understanding matter helps us see why ice melts, water boils, and how gases like oxygen and carbon dioxide are useful in daily life.
- It shows us how tiny particles make up everything around us.
What are the three states of matter?
- There are three states of matter- solid, liquid and gas.
- The state of matter depends on how its particles are arranged and how much energy they have.
Solid:
- In solids, the particles are tightly packed and arranged in fixed positions.
- They can only vibrate but not move freely.
- This is why solids have a definite shape and volume.

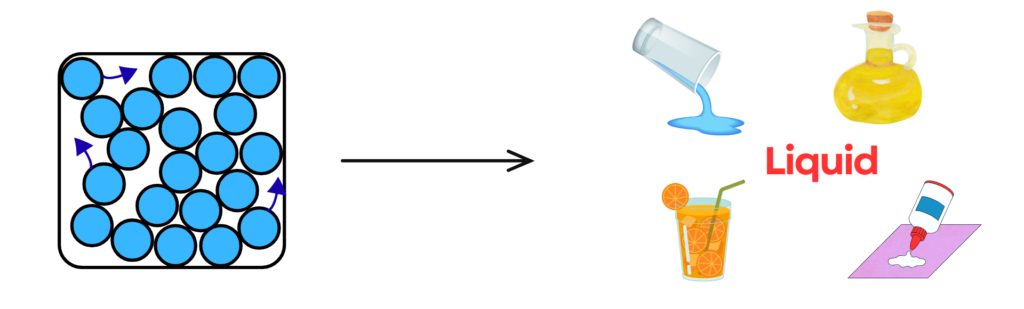
Liquid:
- In liquids, the particles are close together but can move around each other.
- They have a definite volume but no fixed shape, so they take the shape of their container.
Gas:
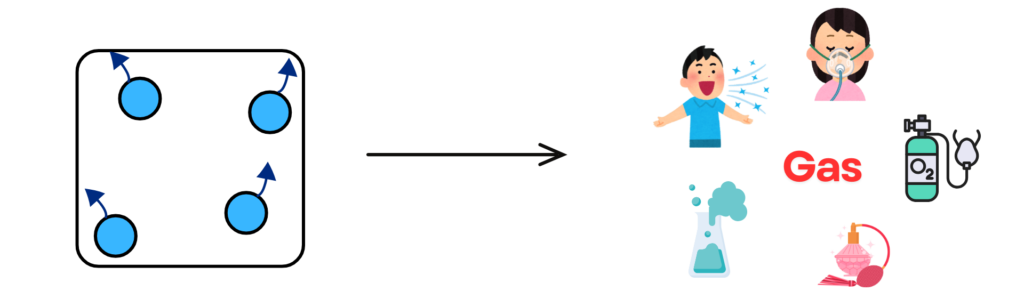
- In gases, the particles are far apart and move quickly in all directions.
- Gases have no fixed shape or volume and will spread out to fill any space available.
What is the Difference between Physical & Chemical Changes?
- Matter can change in many ways. Sometimes it only changes its form or state, and sometimes it turns into a new substance.
- These changes are called physical and chemical changes.
Physical Change:
- A physical change happens when a substance changes its shape or state, but it’s still the same substance and no new material is made.
- These changes are often called reversible changes because the substance can go back to its original state.
Example:
Chemical Change:
- A chemical change happens when a new substance is formed with different properties from the original one.
- These changes are often called irreversible changes because the substance cannot go back to its original state.
Example:

How Does Matter Change State?
- Matter can change from one state to another when heat is added or taken away.
- These changes are called changes of state, and they are physical changes because no new substance is made.
- When a substance gains heat, its particles move faster and spread out.
- When it loses heat, its particles move slower and come closer together.
Main Changes of State:
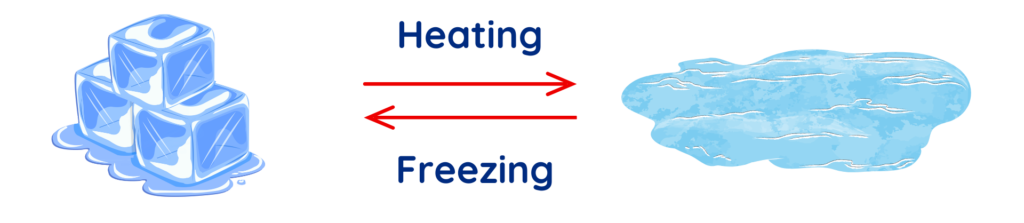
Melting:
- When a solid changes into a liquid by heating.
- Example: Ice melts into water.

Freezing:
- When a liquid changes into a solid by cooling.
- Example: Water freezes into ice.

Evaporation:
- When a liquid changes into a gas by heating.
- Example: Wet clothes dry in the sun because water evaporates.

Condensation:
- When a gas changes into a liquid by cooling.
- Example: Steam condenses into water droplets.

Sublimation:
- When a solid changes directly into a gas without becoming a liquid.
- Example: Dry ice changing into carbon dioxide gas.


What is the Particle Theory & How Does it Explain Energy Changes?
- The particle theory explains how substances behave by describing how their particles move, arrange, and gain or lose energy.
- It helps us understand why matter exists in different states and how it changes when heat is added or removed.
Main idea of theory:
- All matter is made up of tiny particles.
- Particles are always moving — fastest in gases and slowest in solids.
- There are spaces between particles.
- Forces of attraction exist between particles — strong in solids and weak in gases.
- Heating makes particles move faster and spread apart.
- Cooling makes particles move slower and come closer together.
Explanation:
- When a solid is heated, its particles gain energy, vibrate faster, and break free, causing it to melt into a liquid. With more heat, the particles move faster and spread apart, and the liquid boils into a gas.
- When the substance is cooled, the process reverses — particles lose energy, move closer together, and change from-

- The particle theory explains these changes of state through energy transfer and particle movement, helping us understand everything from melting ice in daily life to liquefying gases in industries.

Frequently Asked Questions
Solution:
Matter is anything that has mass and takes up space. Everything around us — like air, water, and metals — is made of matter.
Solution:
- There are three main states of matter — solid, liquid, and gas.
- They differ in how their particles are arranged and move: solids have tightly packed particles, liquids have loosely packed ones that move around, and gases have particles far apart moving freely.
Solution:
- A physical change only changes the form or state of a substance, and no new substance is made — for example, ice melting into water.
- A chemical change makes a new substance with different properties, such as burning wood or rusting iron.
Solution:
- Matter changes state when energy (usually heat) is added or removed.
- Heating makes particles move faster and spread apart, while cooling makes them slow down and come closer together.
Solution:
- The particle theory explains how substances behave by describing the movement, arrangement, and energy of their particles.
- It shows why matter exists in different states and how it changes with energy.
Solution:
- When matter is heated, particles gain energy, move faster, and may break apart to change state (like solid → liquid → gas).
- When it’s cooled, particles lose energy, move closer together, and change back (gas → liquid → solid).
Solution:
- In solids, particles are tightly packed and vibrate in place.
- In liquids, they are close but can slide past each other.
- In gases, they move freely and quickly.
- The more energy particles have, the faster they move.
Solution:
- If the forces between particles are strong, more energy is needed to break them, so the melting and boiling points are higher.
- If the forces are weak, these points are lower.
Solution:
- Below melting point: the substance is a solid.
- Between melting and boiling points: it’s a liquid.
- Above boiling point: it’s a gas.

