Periodic Table – GCSE Chemistry
Introduction
The periodic table is a chart that organizes all known elements in a clear and meaningful way. It helps us understand how elements behave, how they interact, and what properties they share.
- It helps us understand element properties and how different elements are related to each other.
- The periodic table is a powerful tool used by scientists to predict how elements will react, form compounds, or be used in real-life applications.
- It continues to grow and evolve as new elements are discovered and added.
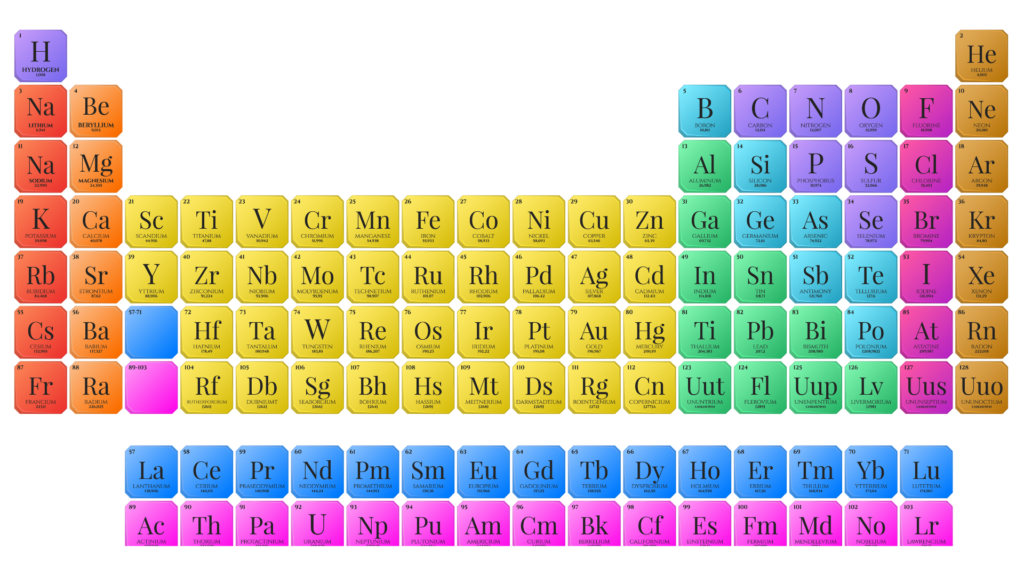
How Mendeleev Expressed His Theory?
Dmitri Ivanovich Mendeleev was a Russian chemist and professor, born on February 8, 1834.
- In 1869, he developed the first version of the periodic table by arranging elements in order of their atomic mass. Mendeleev’s table was unique because he left gaps for unknown elements and predicted their properties in advance.
- His predictions were later proven correct when those missing elements (like gallium and germanium) were discovered.
- Mendeleev is often called the “Father of the Periodic Table” because his work laid the foundation for how we study elements today.

Bold Strategy:
- Unlike others before him, Mendeleev was bold in his approach. If an element did not fit the pattern, he would rearrange it or leave a gap, predicting that an undiscovered element would later fill that position. Remarkably, he not only predicted the existence of several new elements but also described their chemical and physical properties with impressive accuracy.
How He Arranged The Elements?
- Mendeleev arranged elements in order of increasing atomic mass.
- He placed elements with similar properties in the same vertical columns (groups).
- When elements didn’t fit the pattern, he left gaps and predicted undiscovered elements.
- He sometimes rearranged elements to keep similar ones together, even if the mass order was broken.
- His focus was more on chemical properties than just atomic mass.
- This led to a table showing periodic (repeating) patterns in element behavior.
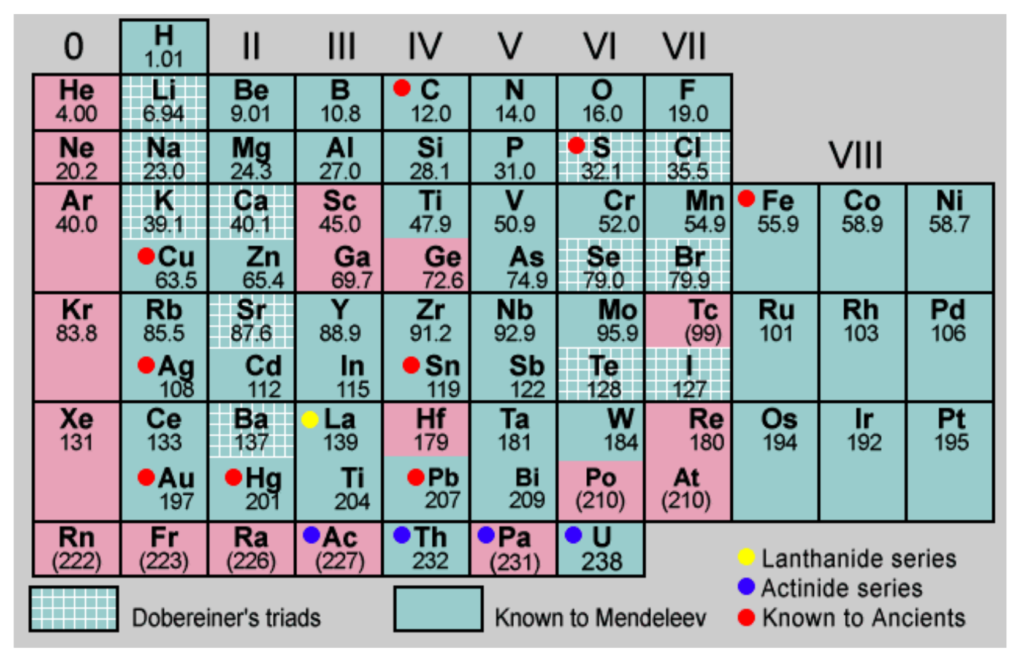
Postulates of Mendeleev’s Periodic Table
- Elements are arranged in order of increasing atomic mass.
- Mendeleev believed that atomic mass was the most important property for organizing elements.
- Elements with similar properties appear at regular intervals.
- This repeating pattern is known as periodicity.
- Elements with similar chemical properties are placed in the same vertical column (group).
- For example, all alkali metals like lithium, sodium, and potassium are in one group.
- The properties of elements are a periodic function of their atomic masses.
- This means that element properties repeat in a predictable way as their mass increases.
- Gaps were left for undiscovered elements.
- Mendeleev left blank spaces in the table and predicted the properties of elements that had not yet been found.
- Incorrect atomic masses were corrected to fit the periodic law.
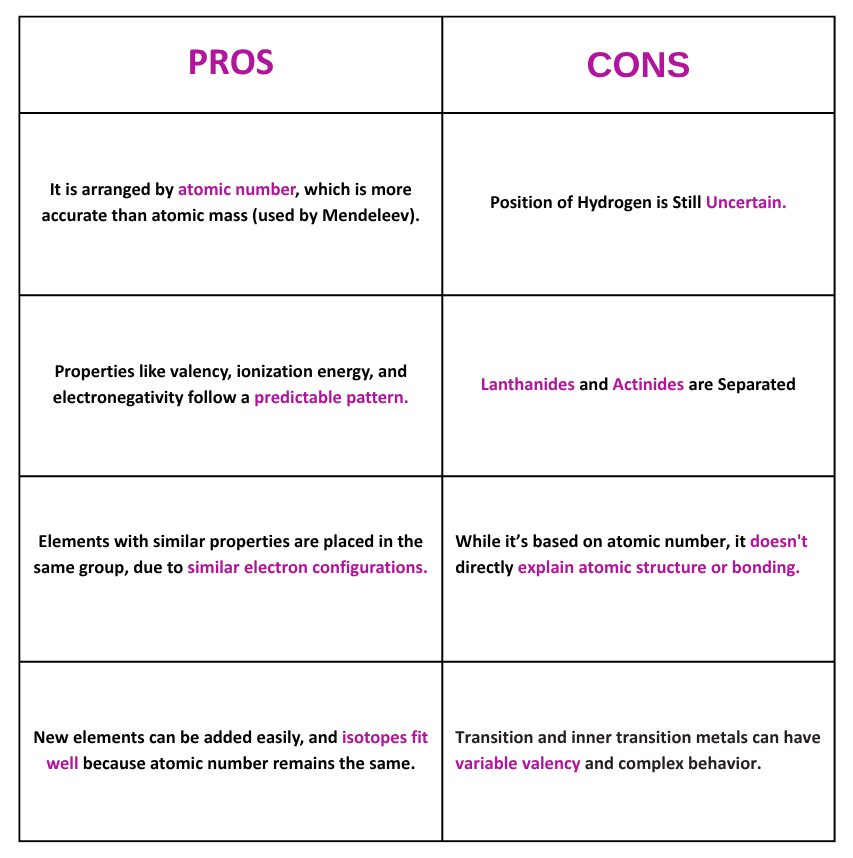
Pros and Cons of Mendeleev’s Periodic Table:

How Modern Periodic Table Get Assembled?
The modern periodic table was introduced by Henry Moseley, a British physicist, in 1913.
- Moseley discovered the concept of atomic number through X-ray experiments.
- He found that each element has a unique number of protons in its nucleus.
- He rearranged Mendeleev’s periodic table by atomic number instead of atomic mass.
- This fixed inconsistencies like the position of iodine and tellurium.

Smart Strategy:
- Henry Moseley arranged elements by increasing atomic number instead of atomic mass. Using X-ray experiments, he discovered that atomic number defines an element’s identity. This fixed errors in Mendeleev’s table and led to the modern periodic law. His strategy gave the periodic table its accurate and current form.
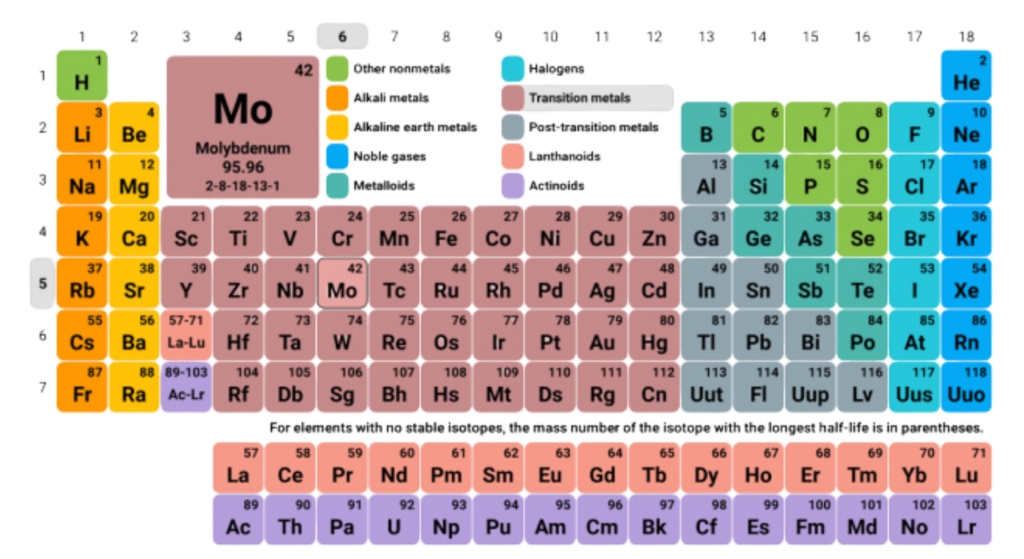
Postulates of Modern Periodic Table
- Elements are arranged in order of increasing atomic number, not atomic mass.
- Properties of elements repeat periodically when elements are arranged by atomic number — this is called periodicity.
- Elements with similar chemical properties are placed in the same vertical groups (columns).
- The table is divided into periods (rows) and groups (columns) based on electron configuration.
- Elements are categorized into s, p, d, and f blocks, depending on the type of orbital their outer electrons occupy.
- Valency and chemical reactivity show a repeating pattern across periods and down groups.
- Metals, nonmetals, and metalloids are grouped based on shared properties and trends (like electronegativity, ionization energy, etc.).
Pros and Cons of Modern Periodic Table:
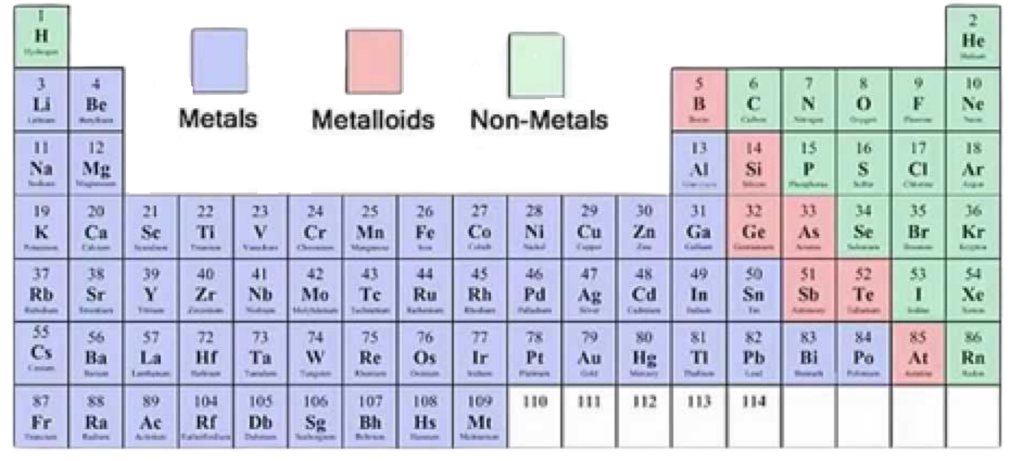
Identification of Elements as Metals and Non-Metals
Position in the Periodic Table:
- Metals are mostly found on the left side and center of the periodic table (Groups 1–12 and part of 13).
- Non-metals are found on the right side of the table (especially Groups 14–18).
- A zig-zag line (starting from Boron to Astatine) separates metals from non-metals; elements along this line are called metalloids.
Physical Properties:
- Metals: Shiny, good conductors of heat and electricity, malleable, and ductile.
- Non-Metals: Dull, poor conductors, brittle, and usually gases or soft solids at room temperature.
Chemical Properties:
- Metals tend to lose electrons and form positive ions (cations).
- Non-Metals tend to gain or share electrons and form negative ions (anions) or covalent bonds.
Examples:
- Metals: Sodium (Na), Iron (Fe), Calcium (Ca), Aluminium (Al)
- Non-Metals: Oxygen (O), Chlorine (Cl), Nitrogen (N), Sulphur (S)
Electronic Configuration of Periodic Elements (1-20)
Electronic configuration is the way in which electrons are arranged around the nucleus of an atom in different energy levels or shells. Each element has a specific number of electrons, and these electrons are filled in shells (orbits) following certain rules:
- The shells are named as K, L, M, N… (starting from the one closest to the nucleus).
- Each shell can hold a Maximum number of electrons:

Rules for Filling Electrons
Aufbau Principle:
- Electrons fill the lowest energy levels (shells) first.
Maximum Electron Rule:
- Each shell has a maximum number of electrons it can hold (as shown above).
Octet Rule:
- Atoms tend to have 8 electrons in their outer shell to be stable (except for Hydrogen and Helium which need 2).
Frequently Asked Questions
Solution:
The periodic table is a chart that arranges all known chemical elements in a specific order based on their atomic number and properties.
Solution:
Dmitri Mendeleev created the first periodic table in 1869 based on atomic mass.
Solution:
- Group: A vertical column (there are 18 groups). Elements in a group have similar properties.
- Period: A horizontal row (there are 7 periods). Properties change gradually across a period.
Solution:
Sodium has 11 electrons → Configuration = 2, 8, 1
Solution:
Use the formula 2n² (where n = shell number):
- K shell (n=1): 2 electrons
- L shell (n=2): 8 electrons
- M shell (n=3): 18 electrons
- N shell (n=4): 32 electrons
Solution:
Elements with 1, 2, or 7 electrons in the outer shell are highly reactive, as they easily lose or gain electrons to become stable.

