Covalent Bonding – GCSE Chemistry
Introduction
- Covalent bonding is a type of chemical bond where two atoms share one or more pairs of electrons to achieve stability.
- This bond typically forms between nonmetal atoms that have similar electronegativities, meaning neither atom can completely transfer electrons to the other (as in ionic bonding.
Example: Water (H₂O) has polar covalent bonds, making it essential for life.
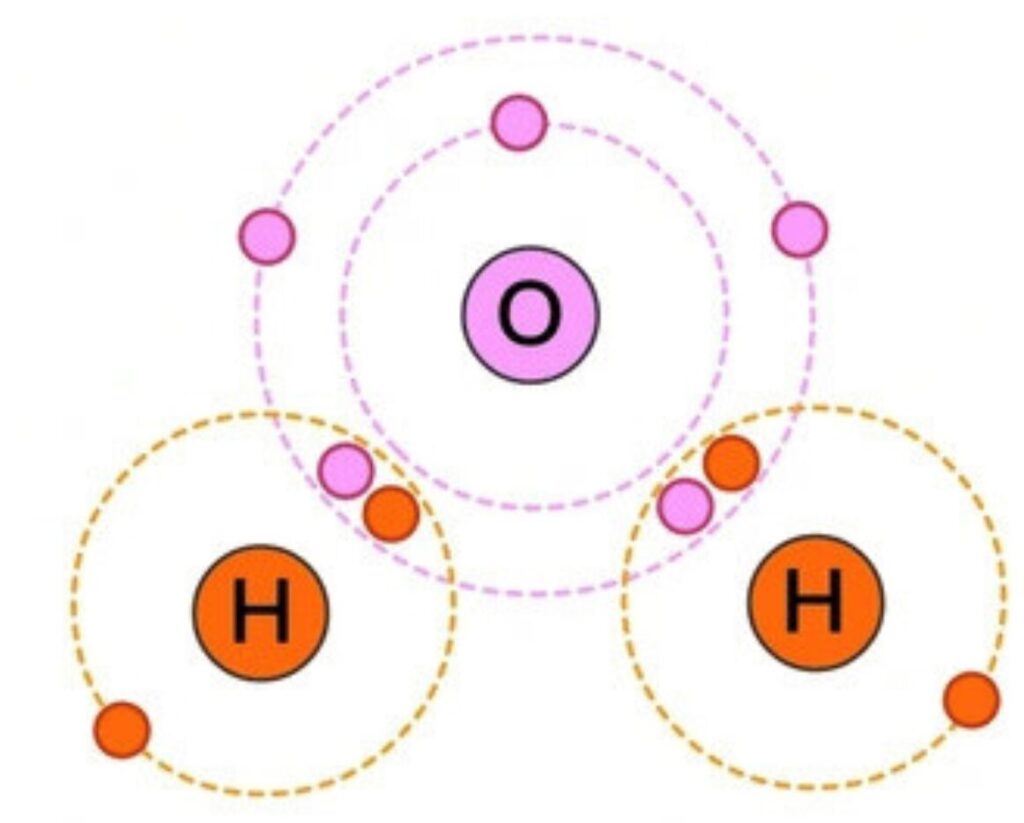
How Covalent Bonds Are Formed?
- Covalent bonds are created when two nonmetal atoms share electrons to complete their outer electron shells. Here’s how it happens:
Step-by-Step Formation:
Step #1: Atoms Approach Each Other
- Two nonmetal atoms (e.g., hydrogen, oxygen, carbon) move close together.
- Each atom has an incomplete outer electron shell and seeks stability.
Step #2: Valence Electrons Interact
- The valence electrons (outermost electrons) of each atom begin to feel the attraction from the other atom’s nucleus.
- Example: Two hydrogen atoms (each with 1 electron) start to share their electrons.
Step #3: Electron Sharing Begins:
- The atoms overlap their atomic orbitals, creating a shared region where electrons move around both nuclei.
- This forms a bonding molecular orbital, where the electrons are most likely to be found.
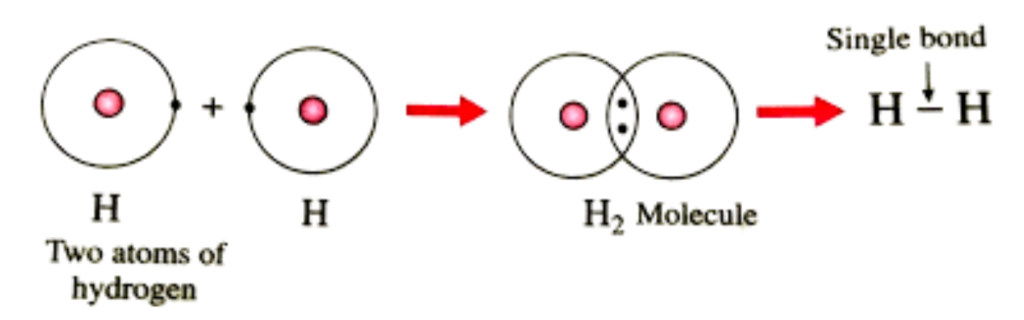
Example: Formation of a Hydrogen Molecule (H₂)
- Two hydrogen atoms (each with 1 electron) approach each other.
- Their 1s orbitals overlap, and the electrons pair up.
- The shared electrons now occupy the space between the nuclei.
- A single covalent bond (H–H) is formed.
Diagrammatically, it can be represented as:
Formation of Covalent Compounds
Occurs Between:
- Non-metal atoms (e.g., H, O, C, N) needing electrons.
Process:
- Atoms share valence electrons to complete their outer shells
- Each shared pair forms one covalent bond
- Can form single (1 pair), double (2 pairs), or triple (3 pairs) bonds
Result:
- Creates discrete molecules (e.g., H₂O, CO₂)
- Molecules have specific 3D shapes (determined by VSEPR theory)
Key Properties:
- Low melting/boiling points
- Poor electrical conductors
- Often gases/liquids at room temperature
Example: Two hydrogen atoms share electrons → H₂ molecule Covalent compounds make up most biological molecules and organic substances.
Types of Covalent Compounds
- Covalent compounds can be systematically categorized based on their structure, bonding characteristics, and physical properties. Here’s an in-depth examination:
Types of Covalent Compounds:
1. Simple Molecular Compounds
Characteristics
- Discrete molecules held by strong intramolecular bonds but weak intermolecular forces
- Typically low melting/boiling points
- Often volatile at room temperature
- Poor electrical conductors
- Subcategories
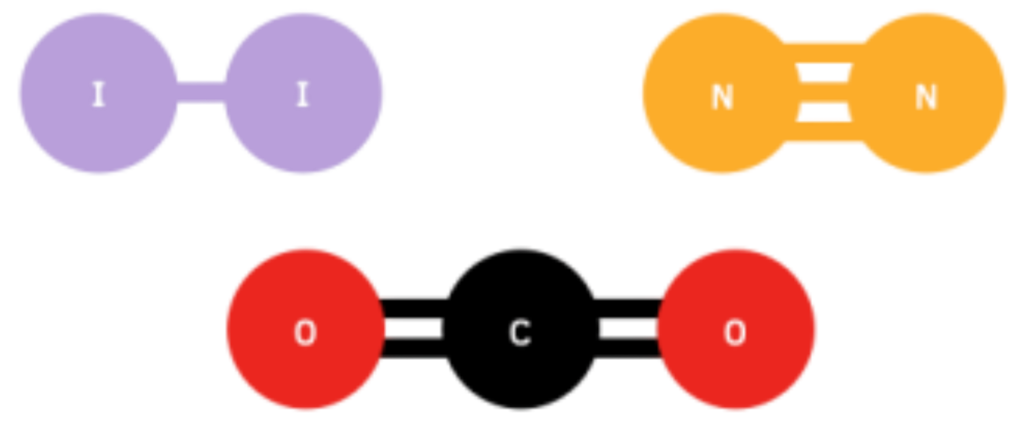
(a) Diatomic Molecules
- Contain exactly two identical atoms
- Examples: N₂ (nitrogen), Cl₂ (chlorine), I₂ (iodine)
(b) Polyatomic Molecules
- Contain three or more atoms
- Examples: H₂O (water) – bent structure, 104.5° bond angle
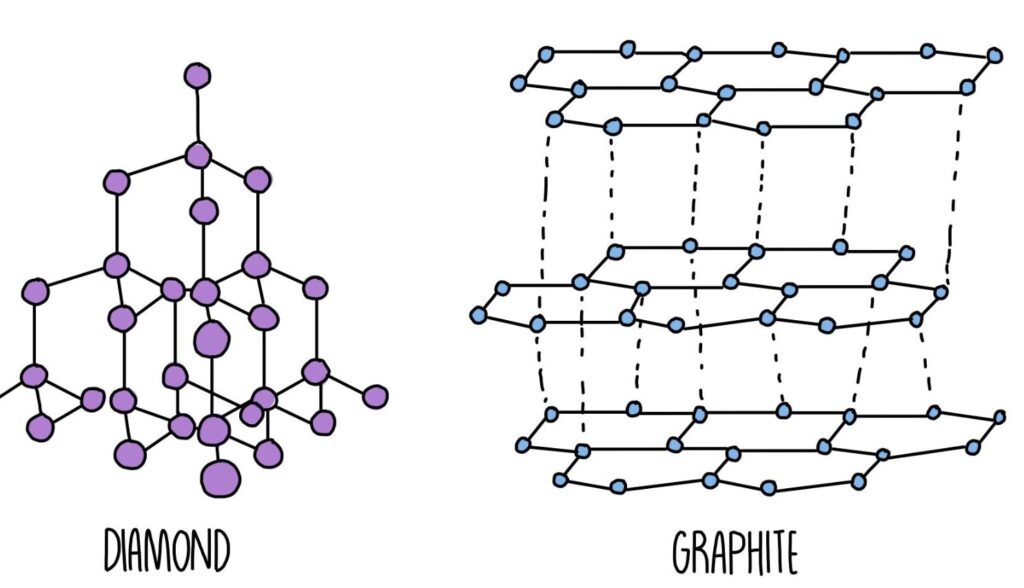
2. Giant Covalent (Macromolecular) Structures
Characteristics
- Three-dimensional network of covalently bonded atoms
- Exceptionally high melting/boiling points
- Generally insoluble in all solvents
- Variable electrical conductivity
Notable Examples:
Diamond
- Each carbon forms 4 tetrahedral bonds
- Hardest known natural material
- Excellent thermal conductor but electrical insulator
Graphite
- Layered
Properties of Covalent Compounds
- Covalent compounds exhibit distinct physical and chemical properties that stem from their molecular structure and bonding characteristics.
1. Physical State
- Molecular Form: Typically exist as gases, liquids, or low-melting solids at room temperature
- Examples: Gases (O₂, CO₂) ,Liquids (H₂O, C₆H₆ benzene), Soft solids (I₂,)
- Network Solids: Exceptionally hard, high-melting materials
- Examples: Diamond (3550°C), silicon carbide (2700°C)
2. Electrical Conductivity
- Poor conductors in all states
- Exception: Graphite (conducts within layers)
- Some become conductive when dissolved (e.g., HCl in water)
- Semiconductors: Special category (Si, GaAs) with tunable conductivity.
3. Solubility
- Poor conductors in all states
- Exception: Graphite (conducts within layers)
- Some become conductive when dissolved (e.g., HCl in water)
- Semiconductors: Special category (Si, GaAs) with tunable conductivity.
4. Isomerism
- Structural isomers: Same formula, different connectivity
- Stereoisomers: Same connectivity, different spatial arrangement
Difference between Ionic And Covalent Bonding

Frequently Asked Questions
Solution:
A covalent compound is formed when two or more nonmetal atoms share electrons to achieve a stable electron configuration (usually an octet). Examples include H₂O (water), CO₂ (carbon dioxide), and CH₄ (methane).
Solution:
Most covalent compounds exist as individual molecules held together by weak intermolecular forces (e.g., van der Waals forces, hydrogen bonds). These forces require less energy to break than ionic bonds.
Solution:
- Polar covalent compounds (e.g., sugar, ethanol) dissolve in water.
- Nonpolar covalent compounds (e.g., oil, methane) do not dissolve in water but dissolve in organic solvents like hexane.
Solution:
Yes! Organic compounds (e.g., methane, ethanol, DNA) are primarily made of C–H and C–C covalent bonds.
Solution:
Diamond: Each carbon is tetrahedrally bonded in a rigid 3D network.
Graphite: Carbon atoms form layers that slide easily due to weak interlayer forces.
Solution:
- Molecule: Any group of bonded atoms (can be elements or compounds).
- Covalent compound: A substance made of molecules with different elements (e.g., H₂O, CO₂).

