Polymers – GCSE Chemistry
Introduction
- Polymers are all around us, found in plastic bags, clothes, non-stick pans, and even in our DNA.
- Many small molecules called monomers join together to form a large molecule known as a polymer.
- In this blog, we’ll explore how polymers are formed, their main types, common uses, and how recycling helps reduce pollution from non-biodegradable plastics.
What is a Polymer?
- A polymer is a large molecule made up of many repeating smaller units called monomers.
- These monomers link together in long chains through strong covalent bonds.
Polymers can be:
Natural:

Synthetic:

What is the Structure of Polymers & How is it Represented?
- Polymers are made of long chains of repeating units called monomers, linked by strong covalent bonds.
- These units are identical in structure.
- Instead of writing the units again and again, they are shown in brackets with lines extending from each side to indicate the continuous chain.
Example:
- Poly(ethene) has the repeating unit:
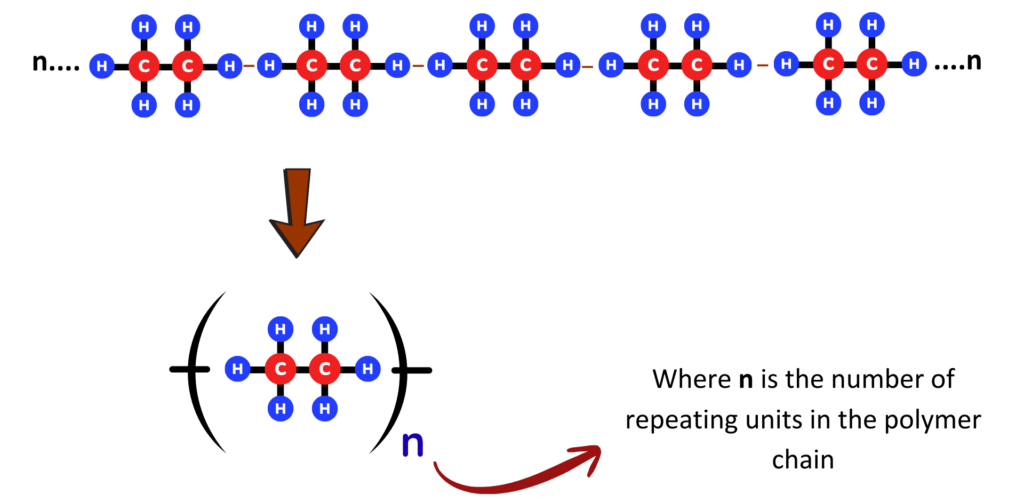
Properties:
- The properties of polymers depend on the chain length and the strength of intermolecular forces.
- Longer chains and stronger forces make polymers tougher, harder, and more durable.
- Shorter chains make polymers softer and more flexible.
Classification of Polymers
Polymers can be classified based on how they are formed and where they come from:
Addition Polymers:
- Addition polymers are formed when many alkene monomers join together.
- So, the double bonds in the monomers break, and the monomers link in a long chain.
- In this reaction, no other molecules are produced.
Example:
- Poly(ethene) is made from ethene (C₂H₄).

Condensation Polymers:
- Condensation polymers are formed when two different monomers, each with two reactive ends, join together.
- Each time the monomers link, a small molecule such as water (H₂O) is released.
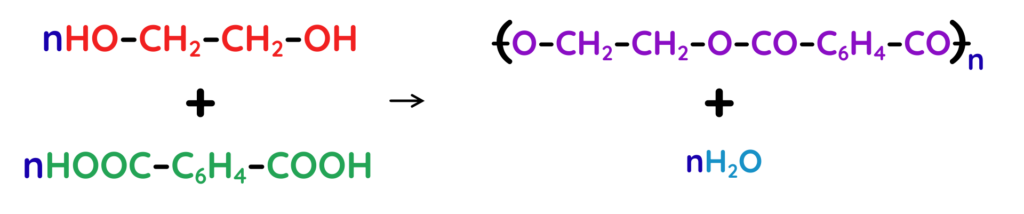
Example:
- Polyester is made from a diol (–OH at both ends) and a dicarboxylic acid (–COOH at both ends). The polymerisation reaction is:
What are the Different Types of Polymers?
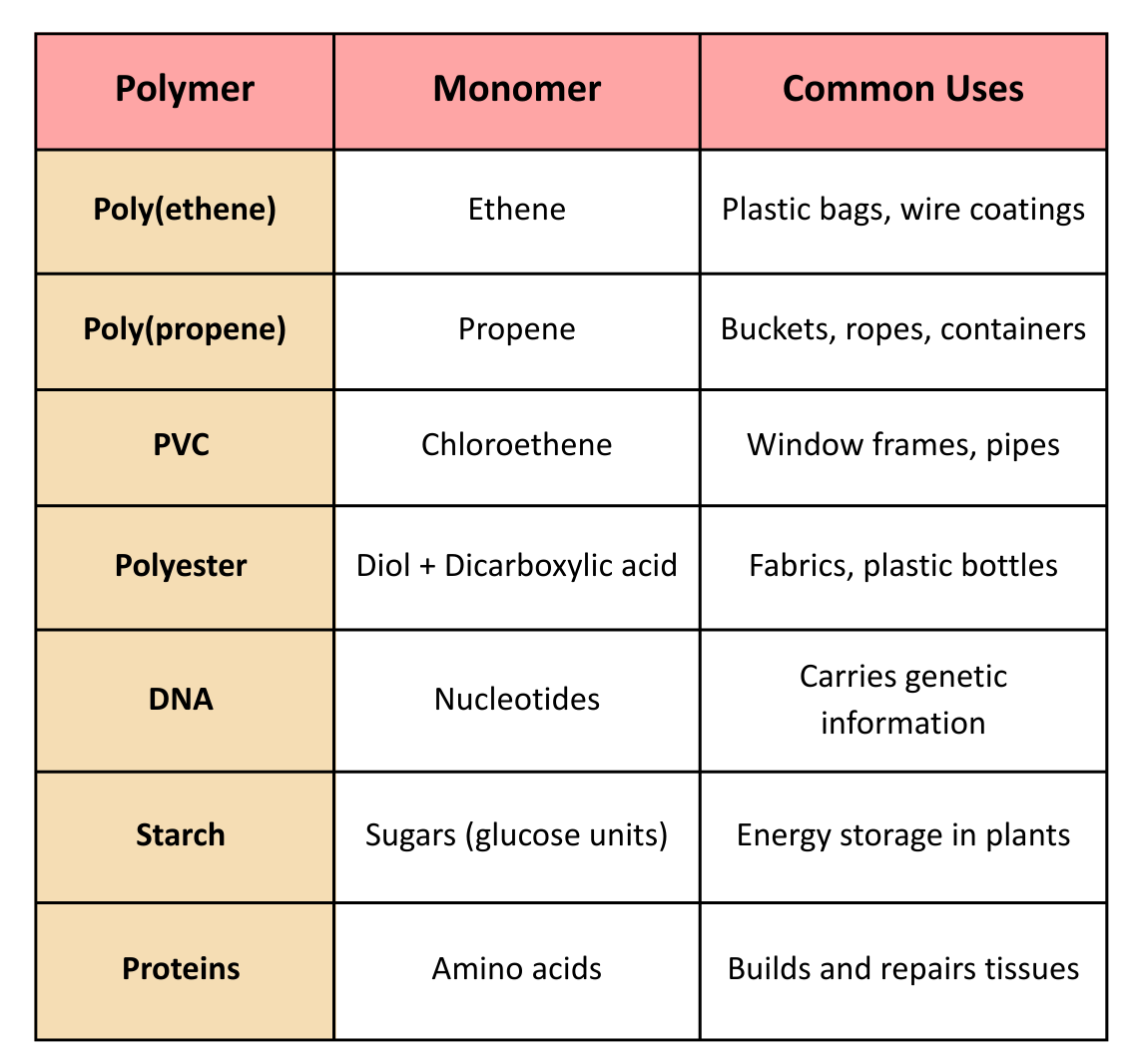
- Polymers come in various types based on the monomers they are formed from and the properties they show.
- Each type has its own distinct features and uses that make it suitable for different purposes in daily life.
Examples:
Uses of Polymers
- Polymers play a major role in our daily lives and in various industries due to their wide range of useful properties such as flexibility, strength, and durability.
- Below are some important examples of how different polymers are used around us:
Poly(propene)
- Poly(propene) is strong and flexible, ideal for containers, ropes, buckets, and packaging.

PVC (Poly(chloroethene))
- PVC (Poly(chloroethene)) is tough and durable, used for window frames, pipes, flooring, and cable insulation.

PTFE (Teflon)
- PTFE (Teflon) is non-stick and heat-resistant, used in cookware, waterproof clothing, and machinery coatings.

Polyesters
- Polyesters are light and strong, used in fabrics, bottles, and packaging films.

Natural Polymers
- Natural polymers like starch, proteins, and DNA are used in food storage, body repair, and genetic information storage.

Frequently Asked Questions
Solution:
A polymer is a large molecule made of many small repeating units called monomers joined together in long chains.
Solution:
Monomers are small molecules, often containing double bonds, that can join together to form polymers.
Solution:
Polymerisation is the chemical reaction where many monomers link together to form a polymer.
Solution:
Addition polymerisation and condensation polymerisation.
Solution:
Alkene monomers with C=C double bonds join to form a polymer, and no other product is formed.
Solution:
They form when two different monomers react together, releasing a small molecule like water each time a bond forms.
Solution:
They differ because of the forces between chains — weak forces make flexible plastics, while strong forces make rigid ones.
Solution:
Polymers are made when many monomers join together; the repeating unit in the polymer has the same atoms as the monomer.
Solution:
They contain strong covalent bonds and are chemically unreactive, so microbes cannot break them down easily.
Solution:
Polymers are used in bags, bottles, pipes, ropes, clothing, coatings, and non-stick cookware.

