Ionic Bonding – GCSE Chemistry
Introduction
- Ionic bonding is a type of chemical bond formed between two atoms when one atom transfers one or more electrons to another atom.
- This transfer of electrons results in the formation of ions—positively charged cations and negatively charged anions. These oppositely charged ions attract each other, creating a strong electrostatic force known as an Ionic Bond.

How Ionic Bonds Are Formed?
- Ionic bonds form when one atom gives up electrons, and another atom takes them. This happens because atoms want to have a full outer shell of electrons (like noble gases) to become stable.
Step-by-Step Formation:
Step#1: Electron Transfer
- A metal atom (like sodium, Na) loses its outer electron(s) because it’s easier to lose than gain.
- A non-metal atom (like chlorine, Cl) gains electron(s) to fill its outer shell.
Step#2: Formation of Ions
- The metal becomes a positive ion (cation) because it loses electrons.
- The non-metal becomes a negative ion (anion) because it gains electrons.
Step#3: Electrostatic Attraction
- The oppositely charged ions attract each other, forming a strong ionic bond.
Example:
- Sodium (Na) has 1 valence electron (easily lost).
- Chlorine (Cl) has 7 valence electrons (needs 1 more).
- Na gives 1 electron to Cl:
- Na → Na⁺ (positively charged)
- Cl + e⁻ → Cl⁻ (negatively charged)
- Na⁺ and Cl⁻ attract, forming NaCl (salt).
Diagrammatically, it can be represented as:

What do you mean by the term ‘ION’?
- An Ion is an electrically charged atom or molecule that forms when an atom gains or loses electrons. Unlike neutral atoms, ions have an unequal number of protons (positive charges) and electrons (negative charges), resulting in a net charge.
How Are Ions Created?
- Ions form through the transfer of electrons between atoms. This happens because atoms strive to achieve a stable electron arrangement, typically resembling the nearest noble gas.
1. Loss of Electrons → Positive Ion (Cation)
- Example: A sodium (Na) atom has 11 protons (+) and 11 electrons (−).
- When it loses 1 electron, it retains 11 protons but only 10 electrons.
- Result: Na⁺ (sodium ion) with a +1 charge.
2. Gain of Electrons → Negative Ion (Anion)
- Example: A chlorine (Cl) atom has 17 protons (+) and 17 electrons (−).
- When it gains 1 electron, it still has 17 protons but now 18 electrons.
- Result: Cl⁻ (chloride ion) with a −1 charge.
Structure of Ionic Compounds as a Lattice Structure
- Ionic compounds form a giant 3D lattice structure due to the strong electrostatic forces between oppositely charged ions. This arrangement maximizes stability by balancing attractions and repulsions.
Key Features of Ionic Lattices
Alternating Ions
- Positive ions (cations, e.g., Na⁺) are surrounded by negative ions (anions, e.g., Cl⁻), and vice versa.
- Example: In NaCl (salt), each Na⁺ ion is surrounded by 6 Cl⁻ ions, and each Cl⁻ is surrounded by 6 Na⁺ ions.
High Melting/Boiling Points
- Strong ionic bonds require large amounts of energy to break, making ionic compounds solid at room temperature.
Brittleness
- When force is applied, like charges may align and repel, causing the lattice to split (e.g., salt shatters when hit).
No Discrete Molecules
- The lattice extends infinitely in all directions, so we write the empirical formula (e.g., NaCl, not “NaCl molecules”).
Real-World Implications
- Solubility: Ionic compounds often dissolve in water because H₂O molecules pull ions apart.
- Applications: Used in batteries (Li-ion), ceramics (MgO), and food preservation (NaCl).
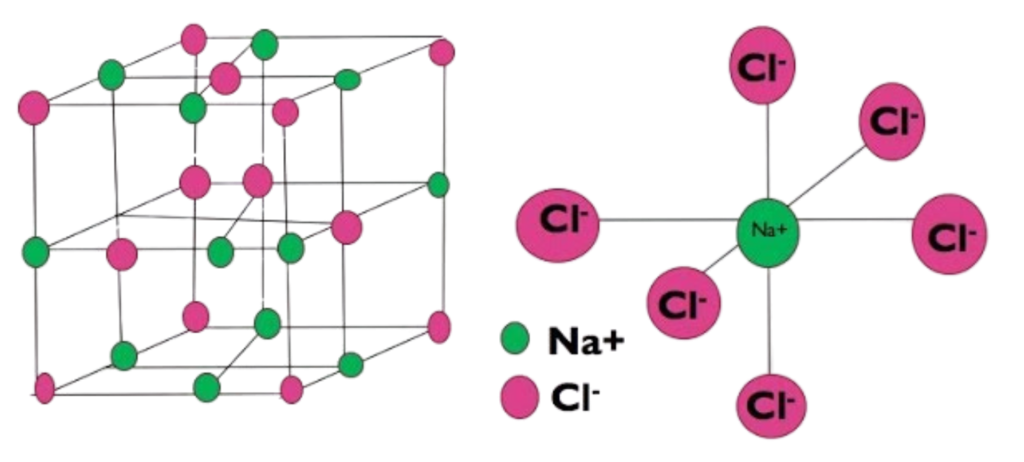
Example: Sodium Chloride (NaCl) Lattice
- Arrangement: Cubic (face-centered).
- Coordination Number: 6:6 (each ion touches 6 oppositely charged ions).
- Visualization: Imagine a 3D chessboard where Na⁺ and Cl⁻ alternate in all directions.
Why Lattice Energy Matters
- Definition: Energy released when gaseous ions form a solid lattice.
- Trends: Smaller ions or higher charges → stronger lattice
Example:
- MgO has a higher melting point than NaCl because Mg²⁺ and O²⁻ attract more strongly than Na⁺ and Cl⁻).
Naming of Ionic Compounds
- Ionic compounds are named systematically based on their cation (positive ion) and anion (negative ion). Here’s how to name them correctly:
Binary Ionic (Metal + Non-Metal)
- Metal name + non-metal root + “-ide”
- Example: NaCl → Sodium chloride
Transition Metals (Variable Charges)
- Metal name + (Roman numeral) + non-metal + “-ide”
- Example: FeCl₃ → Iron(III) chloride
Polyatomic Ions
- Metal name + polyatomic ion name
- Example: NaNO₃ → Sodium nitrate
Hydrated Compounds
- Ionic name + “hydrate” + prefix (e.g., penta-)
- Example: CuSO₄·5H₂O → Copper(II) sulfate pentahydrate
Key Rule: Cation first, anion second.
- Use Roman numerals for transition metals (except Ag⁺, Zn²⁺, Cd²⁺).
- Memorize common polyatomic ions (e.g., SO₄²⁻ = sulfate).
Quick Examples:
- MgO → Magnesium oxide
- Fe₂O₃ → Iron(III) oxide
- NH₄Cl → Ammonium chloride
- CaCO₃ → Calcium carbonate
Difference between Ionic And Covalent Bonding

Frequently Asked Questions
Solution:
An ionic compound is a chemical compound composed of positively charged ions (cations) and negatively charged ions (anions) held together by electrostatic forces (ionic bonds).
Example: Table salt (NaCl) = Na⁺ (cation) + Cl⁻ (anion).
Solution:
Water molecules are polar (have partial charges) and pull ions away from the lattice, dissolving them.
Example: NaCl in water → Na⁺(aq) + Cl⁻(aq).
Solution:
In solids, ions are locked in place in the lattice. When melted/dissolved, ions become mobile and conduct electricity.
Solution:
Most are (e.g., NaCl, CaCO₃), but some are oxides, hydroxides, or other ionic solids (e.g., MgO, NaOH).
Solution:
Salt dissociates into Na⁺ and Cl⁻ ions, which disrupt water’s hydrogen bonding, lowering its freezing point. Sugar (covalent) dissolves but doesn’t split into charged particles, so it’s less effective.
Solution:
Its electrons are too “sticky” (high nuclear charge)—it prefers metallic or covalent bonding.

