Fuels – GCSE Chemistry
Introduction
- Fuels are energy-rich substances that produce heat or power when burned.
- They are essential for running vehicles, cooking food, generating electricity, and more. Common types include solid fuels (like coal), liquid fuels (such as petrol), and gaseous fuels (like LPG).
- Fuels are the foundation of modern life. From lighting homes to powering industries, they are the hidden force that drives progress.

Names and Uses of Fuels
- A Fuel is any substance that can be burned or chemically reacted to produce heat or energy. Fuels are used to power vehicles, machines, generate electricity, and support everyday activities like cooking and heating.
Names and Uses of Common Fuels
- Coal – Used in power plants and industries for electricity and heat.
- Wood – Traditional fuel for cooking and heating in rural areas.
- Petrol (Gasoline) – Fuels cars, scooters, and other light vehicles.
- Diesel – Runs heavy vehicles like trucks, buses, and trains.
- Kerosene – Used in lamps, stoves, and as jet fuel.
- Natural Gas – Powers factories, homes, and electricity plants.
- Biogas – Renewable fuel from organic waste, used for cooking.
- Hydrogen – Clean energy source for fuel cells and space missions.
Important Facts about Fuels
- Fuels are energy sources that release heat or power when burned or reacted.
- Fossil fuels (like coal, petrol, diesel) take millions of years to form from dead plants and animals.
- Over 80% of global energy still comes from fossil fuels.
- Renewable fuels like biogas and ethanol are eco-friendly and reduce carbon emissions.
- Hydrogen and biofuels are future fuels due to their cleaner emissions.
- A good fuel burns easily, gives high energy, and produces less smoke.
Crude Oil Separation
Crude Oil
- Crude oil is a thick, dark-colored liquid found deep underground. It is a natural fossil fuel formed over millions of years from the remains of dead plants and animals buried under layers of rock.
- Crude oil is one of the most important raw materials in the world today.
How is Crude Oil Formed?
- Organic matter (mostly dead microscopic marine organisms) settles at the bottom of ancient seas.
- Over time, it gets buried under layers of mud and rock.
- Heat and pressure gradually transform this material into oil and natural gas.
- The resulting crude oil is trapped in porous rocks, forming underground reservoir.
Uses of Crude Oil
- Crude oil is not used in its raw form. It is sent to refineries, where it is separated into various useful products through a process called Fractional Distillation.
Some of the major products derived from crude oil include:

- Petrol (Gasoline): Used in cars and motorcycles.
- Diesel: Powers trucks, buses, and trains.
- Kerosene: Used in jet engines and for heating.
- LPG (Liquefied Petroleum Gas): Used for cooking and heating.
Fractional Distillation
- Fractional Distillation is a physical separation process used to break down crude oil into useful components, known as fractions, based on their boiling points.
- Crude oil itself is not very useful in raw form, but once separated, it gives us fuels like petrol, diesel, kerosene, and many industrial chemicals.
How Does Fractional Distillation Work?
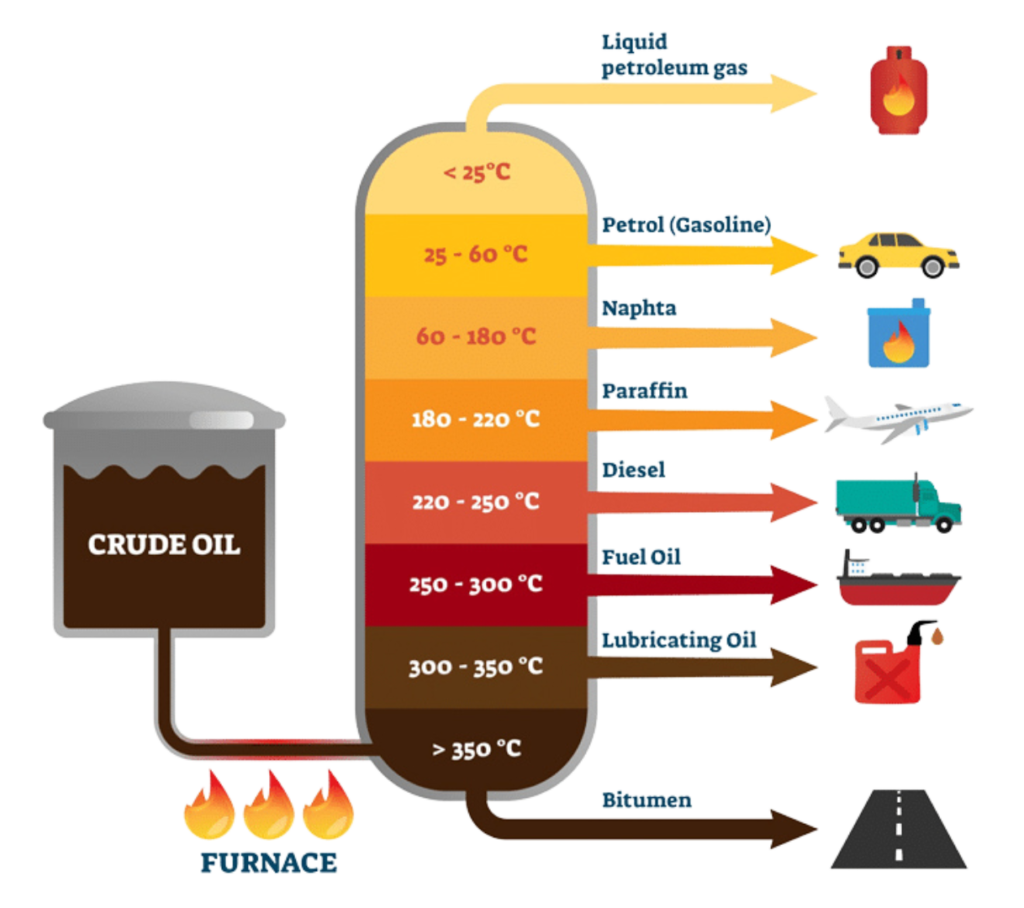
- Heating: Crude oil is first heated in a furnace to around 400°C. It doesn’t boil completely — instead, it becomes a hot mixture of liquid and vapor.
- Entering the Column: The vapor enters the fractionating column, which very tall and has trays at various heights.
- Separation by Boiling Point:
- The column is hot at the bottom and cooler at the top.
- Substances with high boiling points (like bitumen) condense at the bottom.
- Substances with lower boiling points (like gasoline) rise higher before condensing.
- Collection of Fractions: Different products are collected at various levels of the column, depending on where they condense.
Basics of Hydrocarbons and Homologous Series
Basics of Hydrocarbons
- Hydrocarbons are the basic chemical compounds that make up most fuels. They are made of only carbon (C) and hydrogen (H) atoms.
- When hydrocarbons burn in the presence of oxygen, they release a large amount of heat energy, which is why they are widely used as fuels.
Hydrocarbons in fuels are mainly of two kinds:
1. Saturated Hydrocarbons (Alkanes)
- Contain only single bonds between carbon atoms.

- Found in natural gas (e.g., methane, ethane).
- Clean-burning and commonly used in homes and industries.
2. Unsaturated Hydrocarbons (Alkenes and Alkynes)
- Have double or triple bonds between carbon atoms.
- More reactive, used in chemical industries and fuel refining.

Why Are Hydrocarbons Used as Fuels?
- High Energy Content – They release a lot of energy on combustion.
- Abundant in Nature – Found in crude oil, coal, and natural gas.
- Easy to Burn – They ignite easily and burn with a steady flame.
Factors affecting Hydrocarbons
Melting & Boiling Point:
- Fuels with low boiling points (like LPG) vaporize easily and are good for quick ignition. High boiling point fuels (like diesel) need more heat to burn.
Viscosity:
- Fuels with low viscosity (like petrol) flow easily and mix better with air, leading to efficient burning. Thicker fuels may need pre-heating.
Ease of Ignition:
- Fuels with low ignition temperature catch fire easily (e.g., petrol), while those with high ignition temperature (e.g., diesel) are safer but harder to ignite.
Temperature:
- At low temperatures, hydrocarbons may not ignite easily or may burn incompletely, producing harmful gases like carbon monoxide (CO).
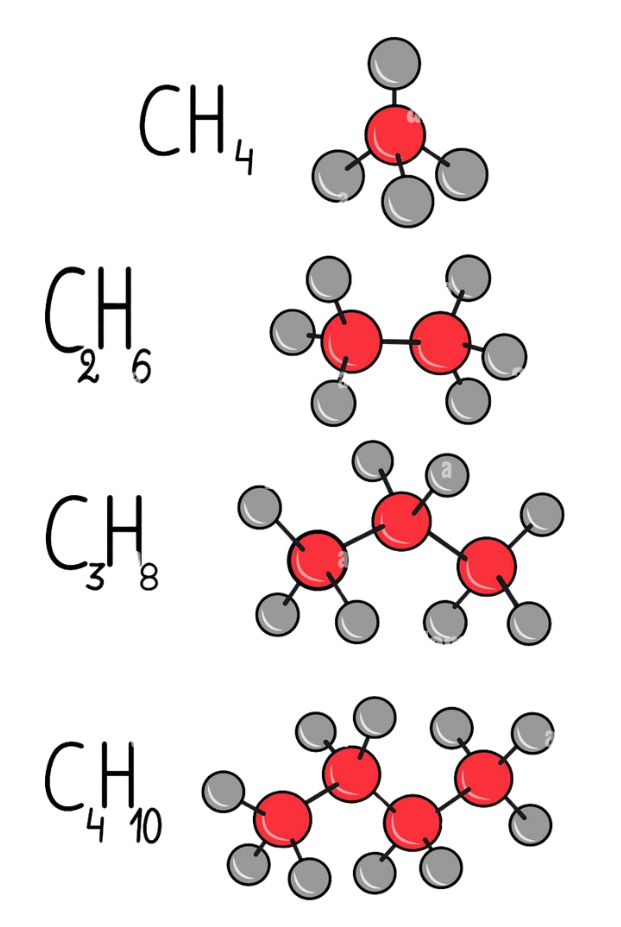
Homologous Series
- A Homologous series is a group of organic compounds with the same functional group and similar chemical properties. Each member differs from the next by a –CH₂– (methylene) group.
Key Features:
- Same general formula (e.g., alkanes: CnH2n+2)
- Gradual change in physical properties.
- Chemical properties remain almost the same
- Each compound differs by 14 u (mass of –CH₂–)
Example: Alkanes Series
Combustion
- Combustion is a chemical process in which a fuel reacts with oxygen to produce heat and light. This process is commonly known as burning. Combustion is the reason fuels like petrol, coal, and LPG are able to release energy when used.
General Equation of Combustion
For a hydrocarbon fuel:

Fuel (Hydrocarbon) + Oxygen → Carbon dioxide + Water + Heat energy
Example:
CH₄ (methane) + 2O₂ → CO₂ + 2H₂O + heat
Importance of Combustion in Fuels
- It is the core process that releases usable energy from fuels.
- Efficient combustion gives more energy and less pollution.
- Incomplete combustion can be dangerous, producing toxic gases like CO.
How to Improve Combustion
- Efficiency Use of clean fuels (like LPG or CNG).
- Ensure adequate oxygen supply.
- Maintain proper air-fuel ratio in engines and burners.
Why Carbon Monoxide acts as a toxic gas?
- Carbon monoxide (CO) is toxic because it interferes with the oxygen-carrying ability of blood.
How CO Affects the Body
- CO binds with hemoglobin (the oxygen-carrying protein in red blood cells) much more strongly than oxygen does.
- This forms a compound called carboxyhemoglobin, which blocks oxygen from reaching the body’s organs and tissues.
- As a result, the brain and heart get less oxygen, leading to serious health problems.
Health Effects of CO Exposure:
- Low exposure: Headache, dizziness, nausea, fatigue.
- High exposure: Confusion, chest pain, unconsciousness, and even death.
- CO is colorless, odorless, and tasteless, so it can go unnoticed — that’s why it’s called a “Silent killer.”
Where CO Comes From:
- Incomplete combustion of fuels (coal, wood, petrol, diesel, LPG) in poorly ventilated spaces.
- Faulty heaters, car exhausts, or gas appliances.
Safety Tip:
- Always ensure good ventilation and use carbon monoxide detectors in closed spaces using fuel-based appliances.
Types of Combustion
Let us understand these Two types of Combustion in detail.
Complete Combustion
- Complete combustion is a chemical reaction in which a fuel burns in the presence of plenty of oxygen, producing only carbon dioxide (CO₂) and water (H₂O) along with heat and light energy.
- It is the most efficient form of burning and is preferred in engines, stoves, and industries because it maximizes energy output and minimizes pollution.
- For any hydrocarbon:
Hydrocarbon + Oxygen → Carbon Dioxide + Water + Heat
Example (Methane):
CH₄ + 2O₂ → CO₂ + 2H₂O + Heat
Conditions for Complete Combustion:
- Adequate oxygen supply
- Proper air–fuel ratio
- Good ventilation or airflow
- Suitable temperature for ignition
Features of Complete Combustion:
- Produces clean, blue flame
- Releases maximum energy
- No harmful gases like carbon monoxide (CO) or soot
- Occurs in gas burners, LPG stoves, well-tuned engines

Advantages:
- Efficient fuel usage
- Less air pollution
- No smoke.
- Safe for health (no CO formation)
- Keeps appliances clean and long-lasting
Incomplete Combustion
- Incomplete combustion occurs when a fuel burns in a limited supply of oxygen, preventing it from fully converting into carbon dioxide and water. Instead, it produces carbon monoxide (CO), soot (carbon particles), and less heat energy.
- It is less efficient and more polluting compared to complete combustion.
- For a hydrocarbon with insufficient oxygen:
Hydrocarbon + Limited Oxygen → CO / C + Water + Less Heat
Example (Methane):
2CH₄ + 3O₂ → 2CO + 4H₂O + Less heat
Conditions for Incomplete Combustion:
- Limited oxygen
- supply Poor ventilation or airflow
- Incorrect air–fuel ratio
- Low burning temperature

Features of Incomplete Combustion:
- Produces a yellow, smoky flame
- Releases less heat energy
- Produces carbon monoxide (a toxic gas)
- May form soot (fine black carbon particles)
- Common in closed spaces or faulty burners
Disadvantages:
- Wastes fuel (less energy per unit)
- Produces toxic gases (like CO)
- Causes air pollution and health hazards
- Soot can damage engines and block chimneys
- Increases maintenance needs for appliances
Conclusion:
- Complete combustion is clean and efficient, producing more energy with fewer pollutants. Incomplete combustion happens with limited oxygen, leading to harmful gases like carbon monoxide and soot. Ensuring good oxygen supply helps in safe and effective fuel use.
Problems caused by Incomplete Combustion
Problems caused by Incomplete Combustion
Produces carbon monoxide (CO):
- A poisonous gas that can cause headaches, dizziness, breathing issues, or even death.
Releases soot (carbon particles):
- Pollutes the air and causes respiratory problems when inhaled.
Wastes fuel:
- Less heat is produced, making the process inefficient and increasing fuel consumption.
Damages appliances:
- Soot can clog chimneys, engines, and burners, leading to poor performance and more maintenance.
Contributes to pollution:
- Causes air pollution, smog, and contributes to climate change due to the release of harmful gases and particles.
Conclusion:
- Incomplete combustion not only reduces fuel efficiency but also poses serious threats to human health, the environment, and mechanical systems.
- The release of toxic gases like carbon monoxide and harmful soot particles highlights the importance of ensuring proper ventilation and oxygen supply during fuel burning to promote cleaner and safer combustion.
Concept of Acid Rain and Pollutants
- Acid Rain refers to any form of precipitation—rain, snow, fog, or even dust—that has been made more acidic than normal due to the presence of sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) in the atmosphere.
- It typically has a pH lower than 5.6, making it harmful to the environment, living organisms, and infrastructure.
How Acid Rain Happens:
- Burning of fossil fuels (like coal, oil, petrol, and diesel) in power plants, factories, and vehicles releases sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) into the air.
- These gases rise into the atmosphere and react with water vapor, oxygen, and other chemicals to form sulphuric acid (H₂SO₄) and nitric acid (HNO₃).
- These acids mix with clouds or moisture in the air.
- Eventually, they fall back to the earth as acidic precipitation — known as “Acid Rain”.
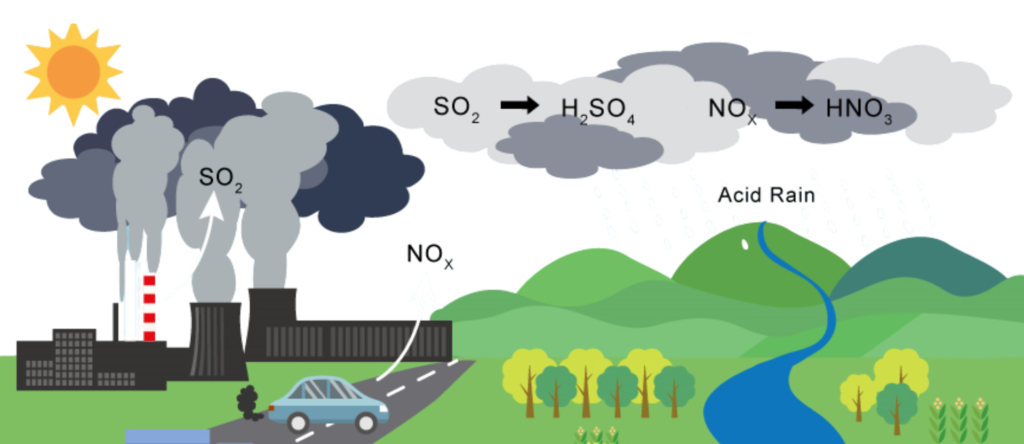
Main Causes of Acid Rain:
Industrial Emissions
- Factories and power stations that burn coal or oil release large amounts of SO₂ and NOₓ.
Vehicle Exhaust
- Cars, buses, and trucks emit nitrogen oxides through the burning of petrol and diesel.
Burning of Biomass
- Burning wood, crop residue, or forest fires also releases acidic gases.
Oil Refineries and Mining
- Processes involving sulfur-containing materials contribute significantly to SO₂ emissions.
Solution To Acid Rain
To reduce acid rain, we must lower emissions of sulfur dioxide and nitrogen oxides.
- This can be done by using clean energy sources: installing pollution control devices, promoting eco-friendly transport, and applying lime to affected areas.
- Public awareness and strict environmental laws also play a key role.

Pollutants
- Pollutants are unwanted or harmful substances released into the environment, especially into the air, water, or soil. They can come from natural sources (like volcanoes or forest fires) or human activities (like burning fuels, industrial waste, or vehicle emissions).
- Pollutants degrade the quality of air, water, and land, causing harm to living organisms and ecosystems.

Common Air Pollutants:
- Carbon monoxide (CO): Toxic gas from incomplete combustion
- Sulfur dioxide (SO₂): From burning coal and oil
- Nitrogen oxides (NOₓ): From vehicle and industrial exhaust.
Solutions to reduce Pollutant particles around us:
- Pollutants can be prevented by using clean energy, reducing vehicle emissions, planting more trees, and avoiding the burning of fossil fuels and waste. Using public transport, recycling, and following pollution control laws also help keep the environment clean.
Cracking and its Significance
- Cracking is a chemical process used to break down large hydrocarbon molecules (usually from crude oil) into smaller, more useful hydrocarbons like petrol, diesel, and gases such as ethene and propene. It can be done using heat (thermal cracking) or heat with catalysts (catalytic cracking).
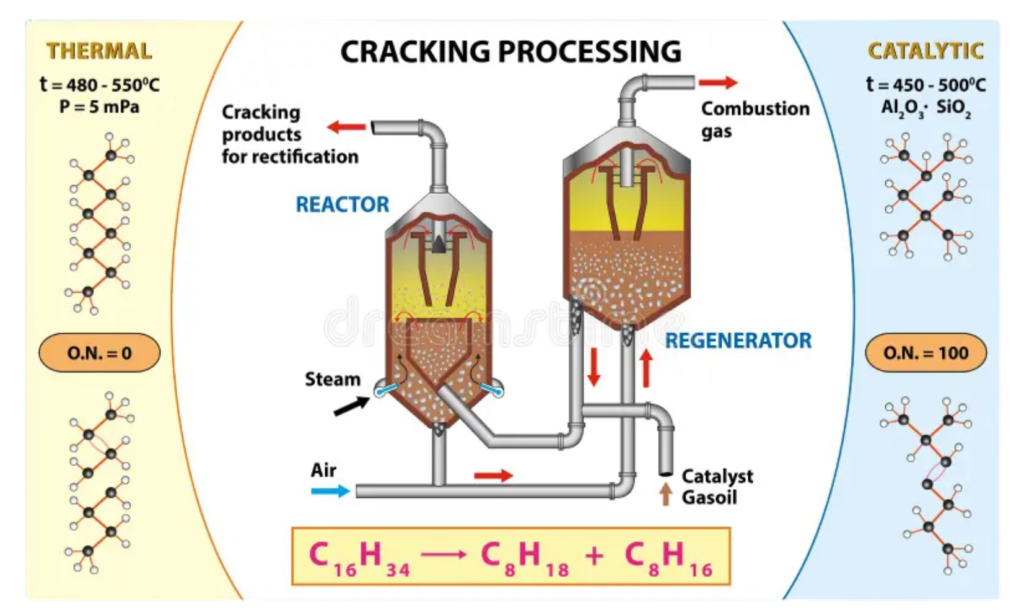
Significance of Cracking:
Increases fuel supply:
- Converts heavier, less useful fractions into high-demand fuels like petrol.
Produces useful gases:
- Makes alkenes (like ethene), which are raw materials for making plastics.
Reduces waste:
- Utilizes heavier oils that would otherwise have limited use.
Supports industry:
- Supplies feedstock for petrochemicals and synthetic materials.
Frequently Asked Questions
Solution:
Renewable fuels can be naturally replenished (like biogas or wood), while non-renewable fuels (like coal and petrol) are formed over millions of years and cannot be replaced quickly.
Solution:
A good fuel should be easily available, affordable, produce high energy, burn cleanly, and be easy to store and transport.
Solution:
Cracking is a process that breaks large hydrocarbons into smaller, more useful ones, such as petrol and ethene.
Solution:
Advantages: Hydrogen is clean (produces only water), renewable, and gives more energy per unit mass. It helps reduce air pollution and greenhouse gases.
Disadvantages: Hydrogen is hard to store, expensive to produce, needs special infrastructure, and is highly flammable, which raises safety concerns.
Solution:
Burning fossil fuels releases sulfur dioxide (SO₂) and nitrogen oxides (NOₓ), which cause acid rain when mixed with rainwater.
Solution:
Yes, all members of a homologous series show similar chemical behavior due to the same functional group present in them.

