Electrical Circuit – GCSE Physics
Introduction
- An Electrical circuit is a closed path that allows electric current to flow through it.
- It connects electrical components using conductors to perform a specific function using electricity.
- Electrical circuits make it possible to control and distribute electrical energy safely and efficiently.
Where it is used:

What is an Electrical Circuit?
- An Electrical circuit is a closed loop path that allows electric current to flow, allowing energy to power devices and systems.
- It connects some components so that electricity can do useful work.
- Without electrical circuits, we could not use electricity to light our homes, charge devices, run machines, or operate communication systems.
Basic Electric Circuit:
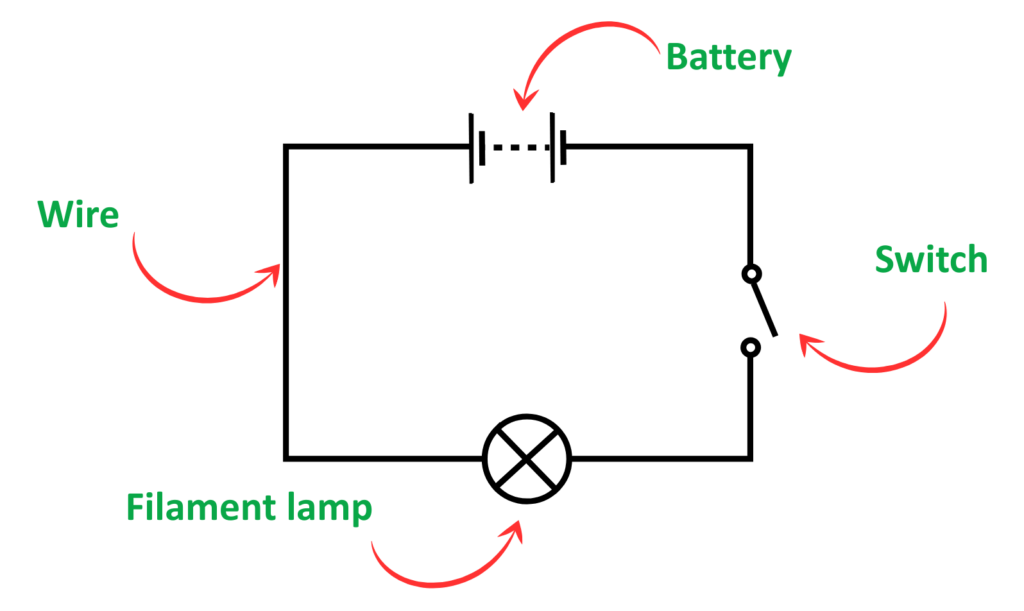
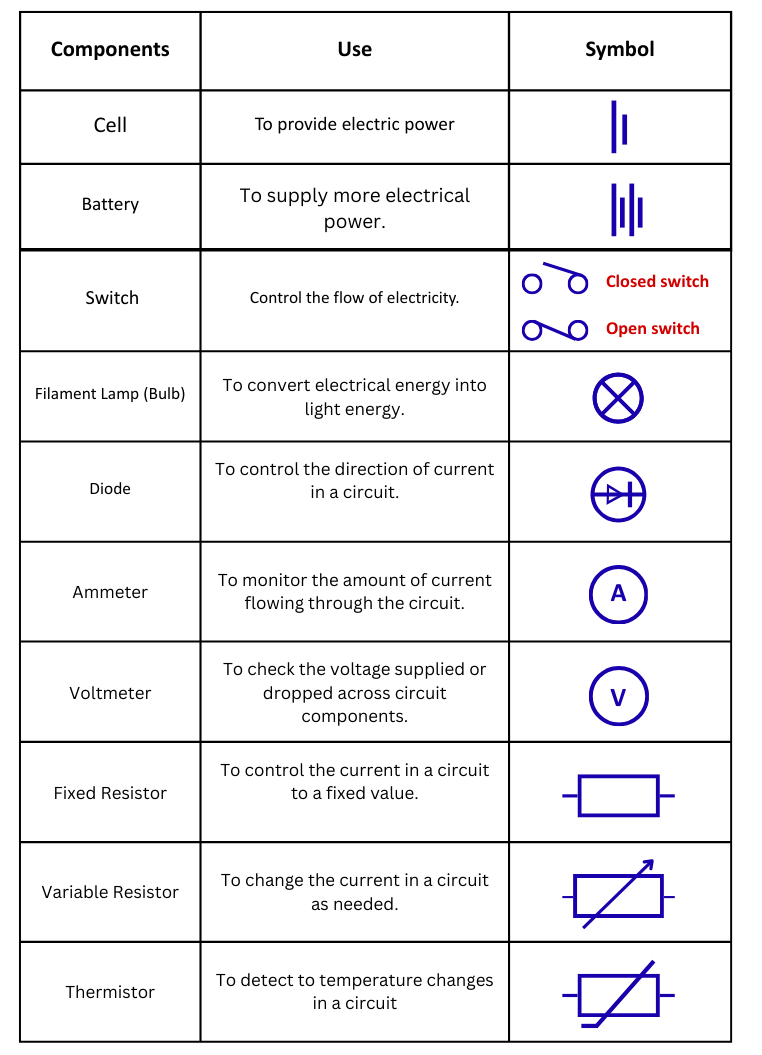
Components used in an electrical circuit with their symbols:
What are Series and Parallel Circuits?
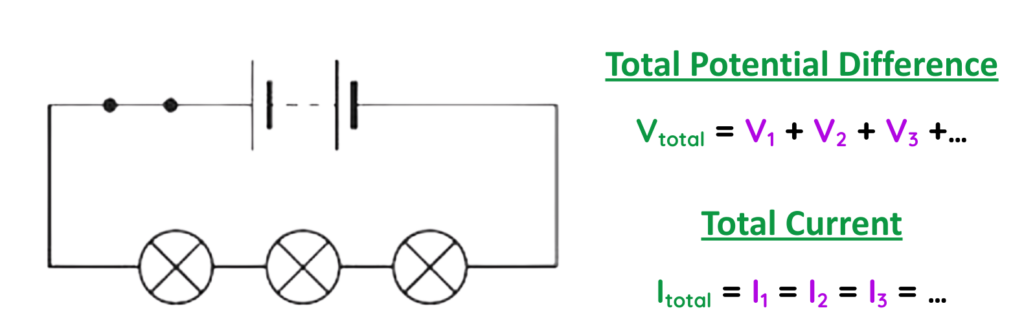
Series Circuit
- In Series circuit, the components are connected end-to-end in a single path, so the same current flows through all of them.
Example:
- A simple flashlight with two batteries.
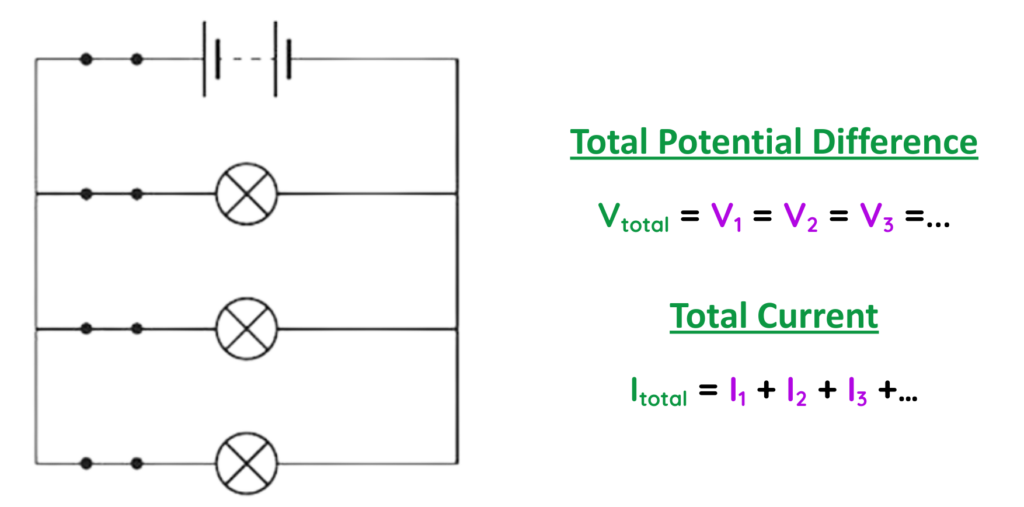
Parallel Circuit:
- In Parallel circuit, the components are connected across multiple paths, so voltage is the same across each branch, but current can vary.
Example:
- Home appliances (lights, fans, etc.) wired separately to the mains.
What is Potential Difference, Current & Resistance?
Potential Difference:
- Definition: The force required for the flow of electrons in a circuit is called potential difference.
- Unit: Volts (V)
- Provided by: A cell or battery.
Current:
- Definition: The flow of electric charge (usually electrons) through a conductor is called current.
- Unit: Amperes (A)
- Provided by: A cell or battery pushing charges through the circuit.
Resistance:
- Definition: The property of a material that opposes the flow of electric current.
- Unit: Ohms(Ω)
- Provided by: Resistors and the materials of wires/components in the circuit.
What is Ohm’s Law?
- Ohm’s Law states that the current through a conductor is directly proportional to the potential difference across it, if the temperature remains constant.
- It is written as:

Where:
- V = Potential difference
- I = Current
- R = Resistance
 Solved Example:
Solved Example:
Problem: If a circuit has a current of 3 A and a resistance of 3 Ω, find the voltage across it.
Solution:
Step #1: Given
- A = 3A
- R = 3Ω
Step #2: Using formula

Step #3: Plug the values

Therefore, the voltage across the circuit is 9V.
Final Answer: 9V
How are Charge, Current & Time Related?
Charge
- Definition: A measure of the total current that flowed within a certain period of time. It is carried by particles like electrons and protons.
- Symbol: Q or q
- Unit: Coulomb (C)
How to Calculate Charge:
- Formula:

Where,
- Q = Charge
- I = Current
- t = Time
 Solved Example:
Solved Example:
Problem: A current of 5 A flows through a circuit for 6 seconds. Calculate the charge that flows through the circuit.
Solution:
Step #1: Given
- A = 5A
- t = 6s
Step #2: Using formula

Step #3: Plug the values
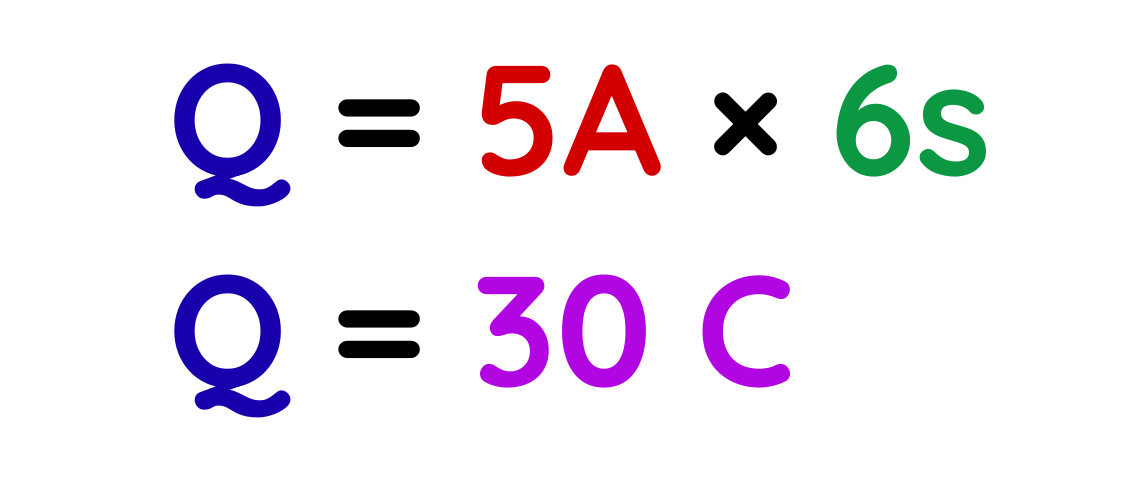
Final Answer: 30 C
How are Energy, Voltage & Charge Related?
Energy
- Definition: An electricity, energy is the work done when charge moves through a voltage.
- Symbol: E
- Unit: Joule (J)
How to Calculate Charge:
- Formula:

Where:
- Q = Charge
- E = Energy
- V = Voltage
 Solved Example:
Solved Example:
Problem: A charge of 15 C moves through a voltage of 9 V. Calculate the energy transferred.
Solution:
Step #1: Given
- Q = 15Q
- V = 9V
Step #2: Using formula

Step #3: Plug the values

Therefore, 135 joules of energy are transferred.
Final Answer: 135 Joules
Solving Questions with Circuit Diagrams
Steps to Solve Question with Circuit Diagram
- Step #1: Identify given values in the circuit.
- Step #2: Choose the correct formula.
- Step #3: Substitute values and calculate the unknown.
 Solved Example:
Solved Example:
Problem: A circuit has a 12 V battery and a 4 Ω resistor in series. Find the current.
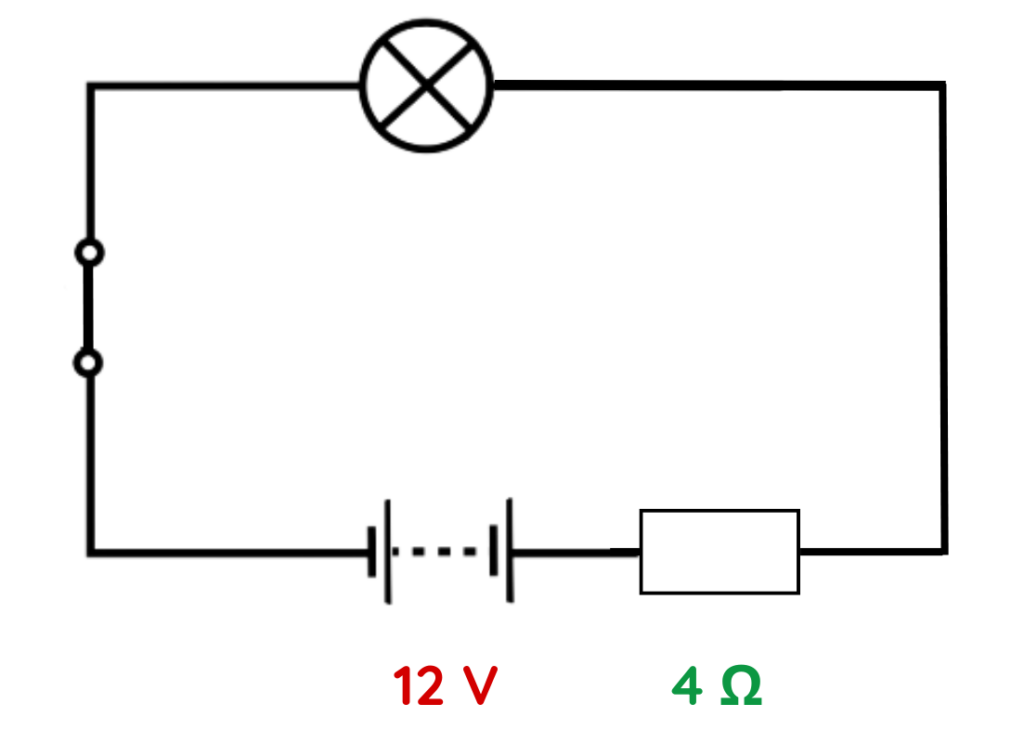
Solution:
Step #1: Identify given values in the circuit.
- V = 12 V
- R = 4Ω
Step #2: Choose the correct formula.
- Using Ohm’s Law:

Step #3: Substitute values and calculate the unknown.
- Substitute the values:
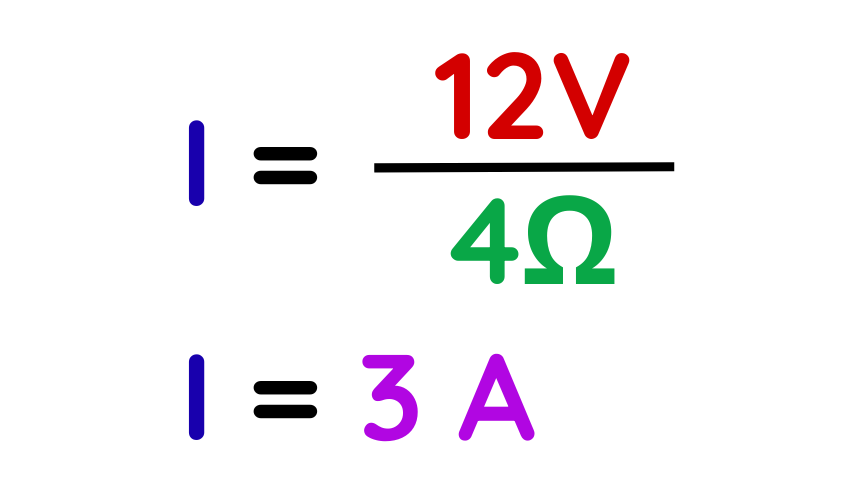
Therefore, the current flowing in the circuit is 3 A.
Final Answer: 3 A
 Solved Example:
Solved Example:
Problem: A parallel circuit has a 10 Ω resistor and a 20 Ω resistor. The current through the 20 Ω resistor is 0.6 A. Calculate:
1. The current through the 10 Ω resistor, and
2. The total voltage of the cell.

Solution:
Step #1: Identify given values in the circuit.
- R1 = 10Ω
- R2 = 20Ω
- I2 = 0.6 A
Step #2: Choose the correct formula.
- In parallel circuits,
- Voltage across each branch is the same:

Step #3: Substitute and calculate.
- The voltage across the resistors:
Using,
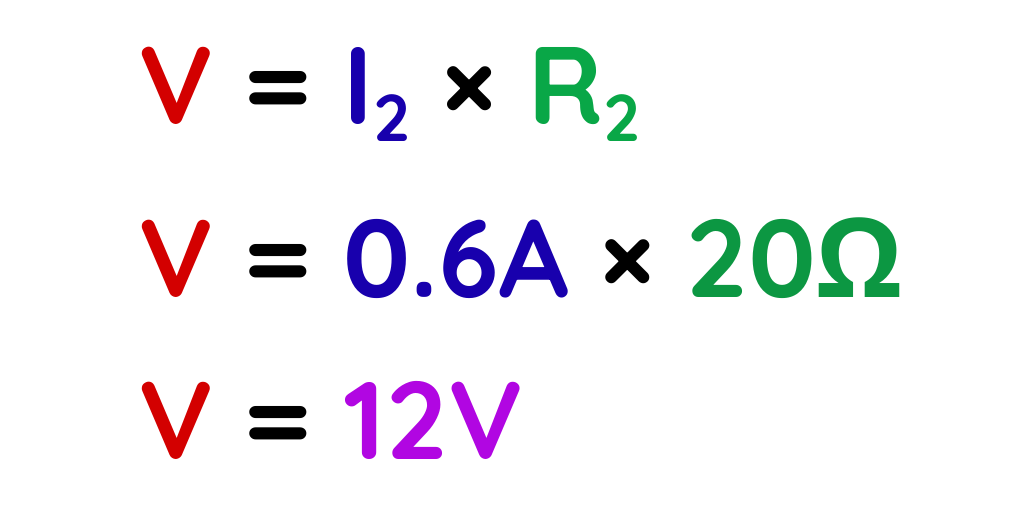
The voltage across the 10 Ω resistor is also 12 V.
- The current through the 10 Ω resistor:
Using,

The current through the 10 Ω resistor is 1.2 A.
- The total current from the cell:
In parallel:

The total current from the cell is 1.8 A.
 Solved Example:
Solved Example:
Problem: A battery of 9 V is connected in series with a 3 Ω resistor. Calculate the charge flowing through the circuit in 4 seconds.
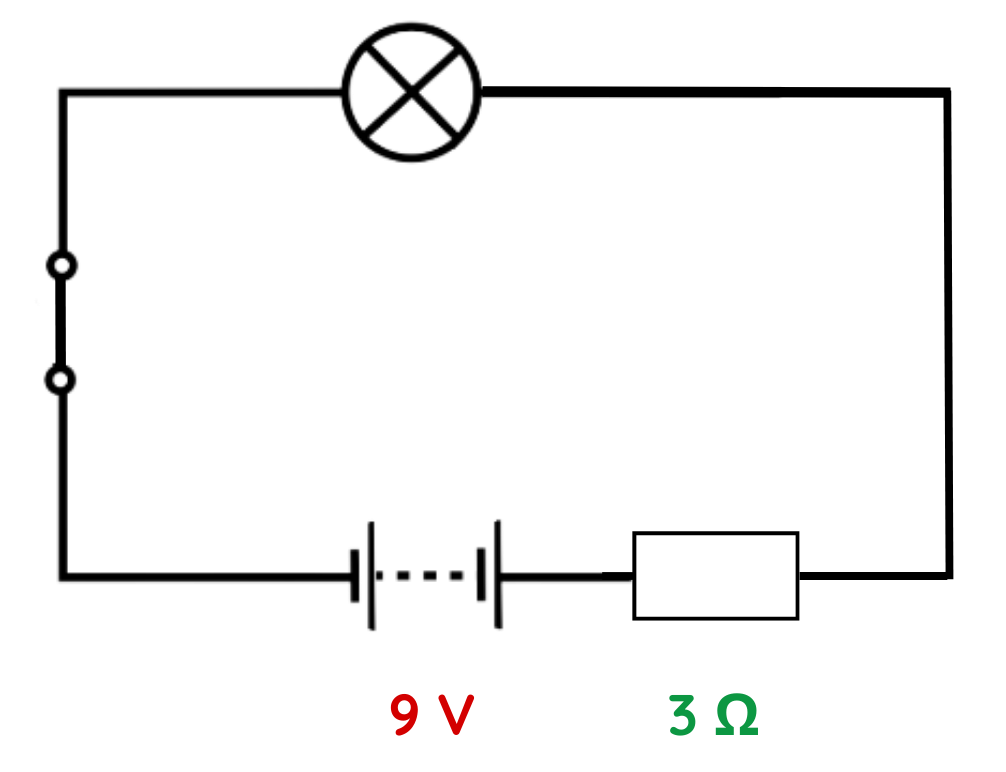
Solution:
Step #1: Identify given values in the circuit.
- V = 12V
- R = 4Ω
- t = 4s
Step #2: Choose the correct formula.
- To find charge (Q), use:

First, find current (I) using Ohm’s Law:
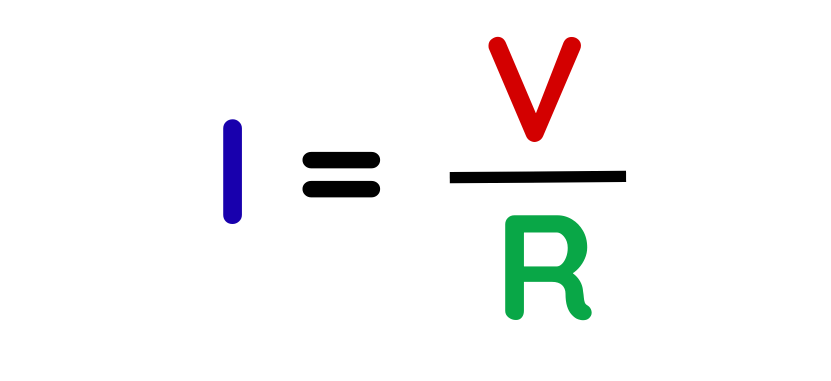
Step #3: Substitute and calculate.
- Find the Current:
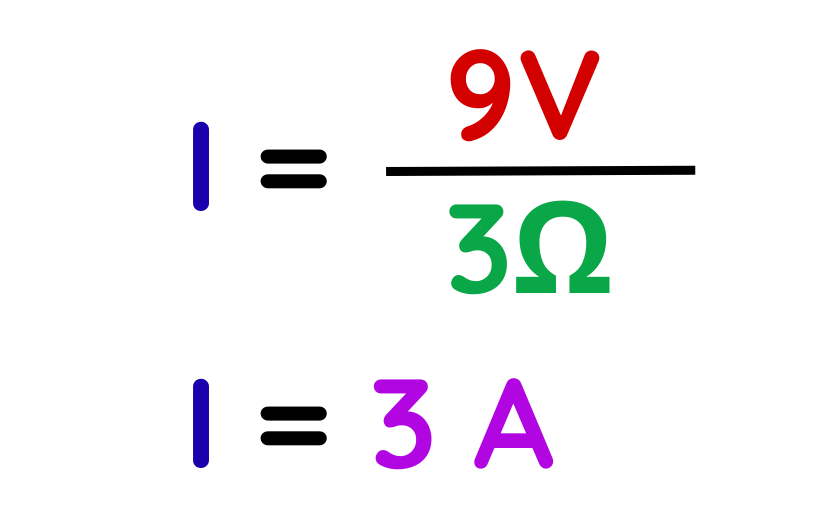
- Find the Charge:

The charge flowing through the circuit in 4 seconds is 12 C.
Final Answer: 12 C
Frequently Asked Questions
It is a closed path that allows electric current to flow.
Ohm’s Law states that:
V = I × R
Electric charge is a property of particles like electrons and protons that causes them to experience a force in an electric field.
It is the work done to move unit charge between two points. Measured in volts (V) using a voltmeter.
It is the opposition to the flow of electric current. Measured in ohms (Ω) using an ohmmeter.
Current stays the same in all parts of the circuit. It has only one path to flow.

Table of Content
Radioactivity Dangers and Medicines – GCSE Physics
Introduction
- Radioactivity means a process in which unstable atom nuclei spontaneously emit energy in order to gain stability.
- Radiations are primarily of three types – Alpha(α) Radiations, Beta(β) Radiations and Gamma(γ) Radiations.
- There are many medicinal applications of Radioactivity. Also, the small amount of radiation is safe, but higher exposure to radiation can damage cells and DNA, causing cancer and serious health issues.

Let us study the applications of radiation in medical treatments, and also know about the dangers of radiation
Uses of Radiation
1. Food Irradiation
- In our environment, some foods naturally contain radioactive substances. We know that food contains a special type of bacteria that cause it to decompose eventually. But some of those bacteria can cause food poisoning.
- To prevent food poisoning, the food is irradiated using gamma rays. Here, the microorganisms are killed.
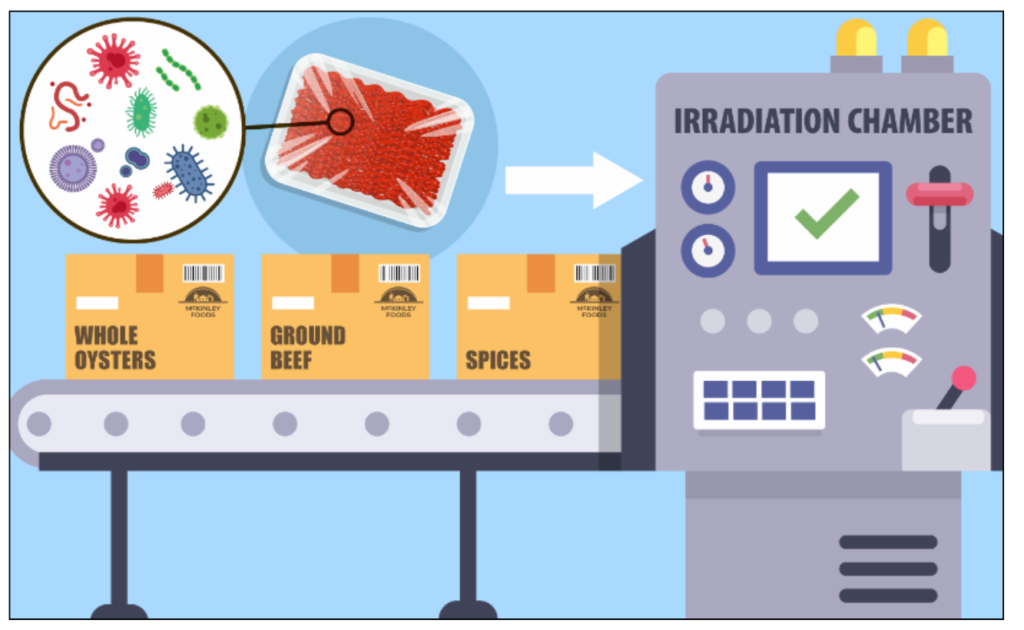
Steps
- Food is typically packaged before irradiation.
- Packaged food is moved through an irradiation chamber.
- The food is exposed to a controlled dose of ionising radiation.
- Radiation disrupts the DNA of microorganisms, preventing them from reproducing.
- Irradiation does not heat the food or leave behind any chemical residue.
2. Radioactivity Detecting
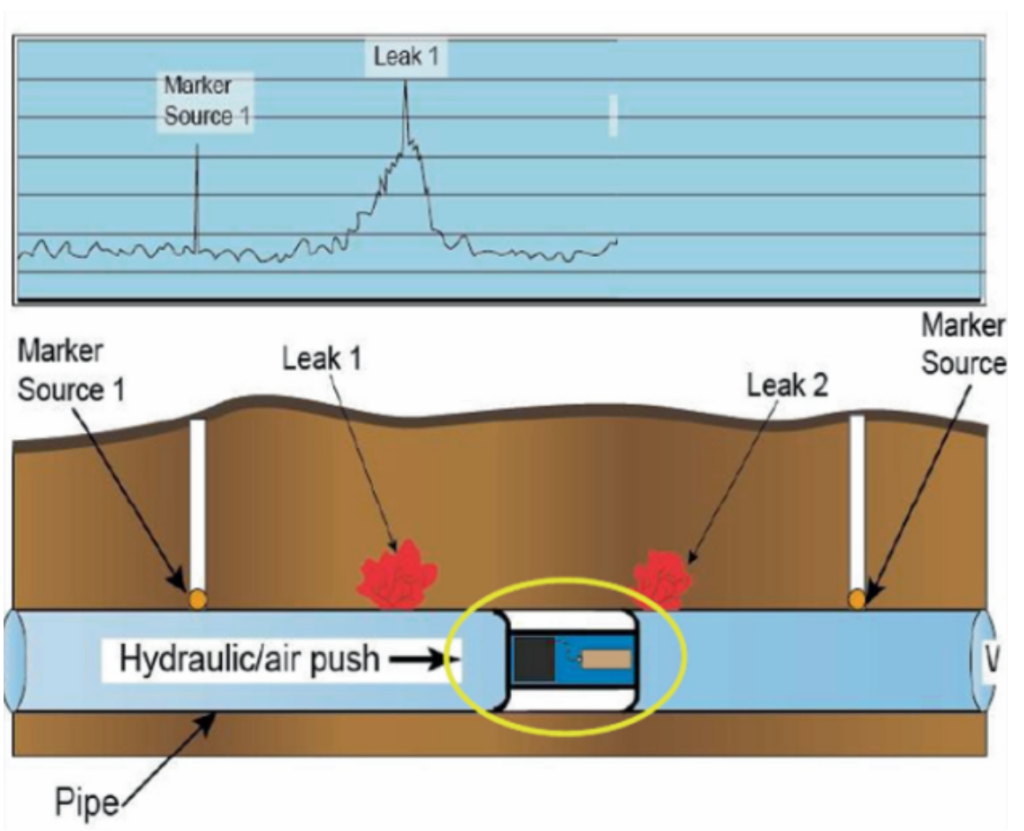
- Radioactive substances are used as tracers. For example, the underground pipe leakage detection by the Geiger-Muller Tube.
Steps
- Sensitive detectors like the Geiger-Muller tube are used to scan the area surrounding the underground pipe.
- The radioactive tracer is introduced into the pipe. The tracer flows through the pipe, and where there is leakage, the tracer-containing fluid will escape.
- The detectors measure the radiation levels. Higher level of readings indicate the presence of radioactive tracers, hence pointing to the pipe leakage location.
3. Smoke Alarms
- Radioactive substances are used as smoke alarms.
Steps
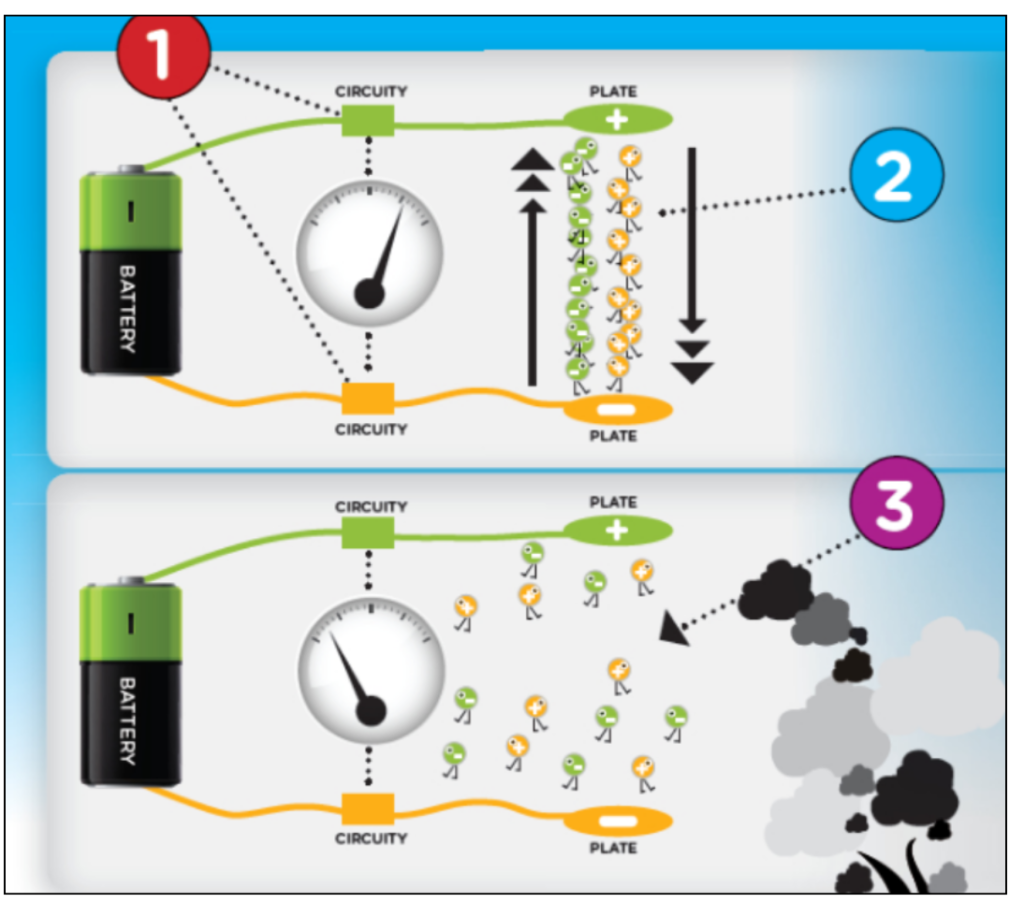
- The Smoke Alarm contains an electrical circuit, which has two electrically charged plates with an air gap between them. A source is connected to the plate, which releases Americium-241.
- This Radioactive substance will ionise the air molecules, now they are attracted to oppositely charged plates of the circuit, allowing a small amount of current to flow that is measured by the detector.
- The Smoke alarm will not sound as long as the current is flowing. But when the smoke gets into the gap between the plates, the ions will slow down, decreasing the amount of current measured by the detector. Now, at this point of current dropping, the alarm will start ringing.
4. Checking Thickness
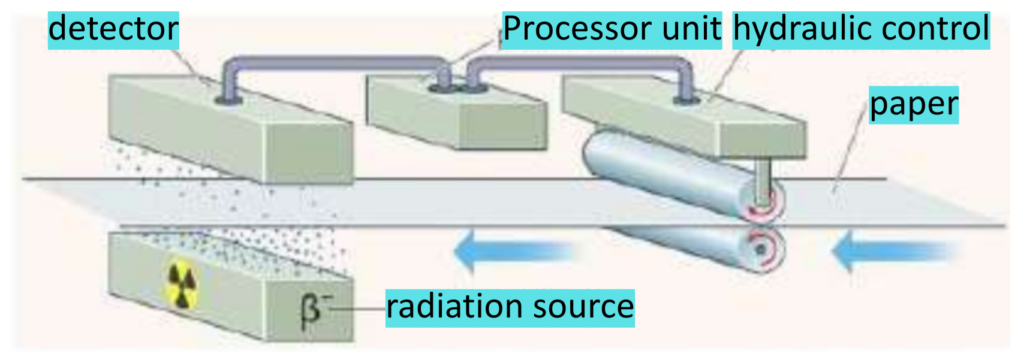
- Paper is made by squeezing wood pulp between two rollers. These rollers must apply same force on wood pulp to squeeze it into the same sized paper.
Steps
- The detector counts the rate at which beta particles get through the paper on one side.
- When the paper is too thin, the count rate detected is higher. Then, a computer senses this rise in count rate and sends a signal to decrease the pressure applied by rollers to maintain the thickness of paper. Similarly, when the paper is thick, the opposite happens.
Dangers of Radiation
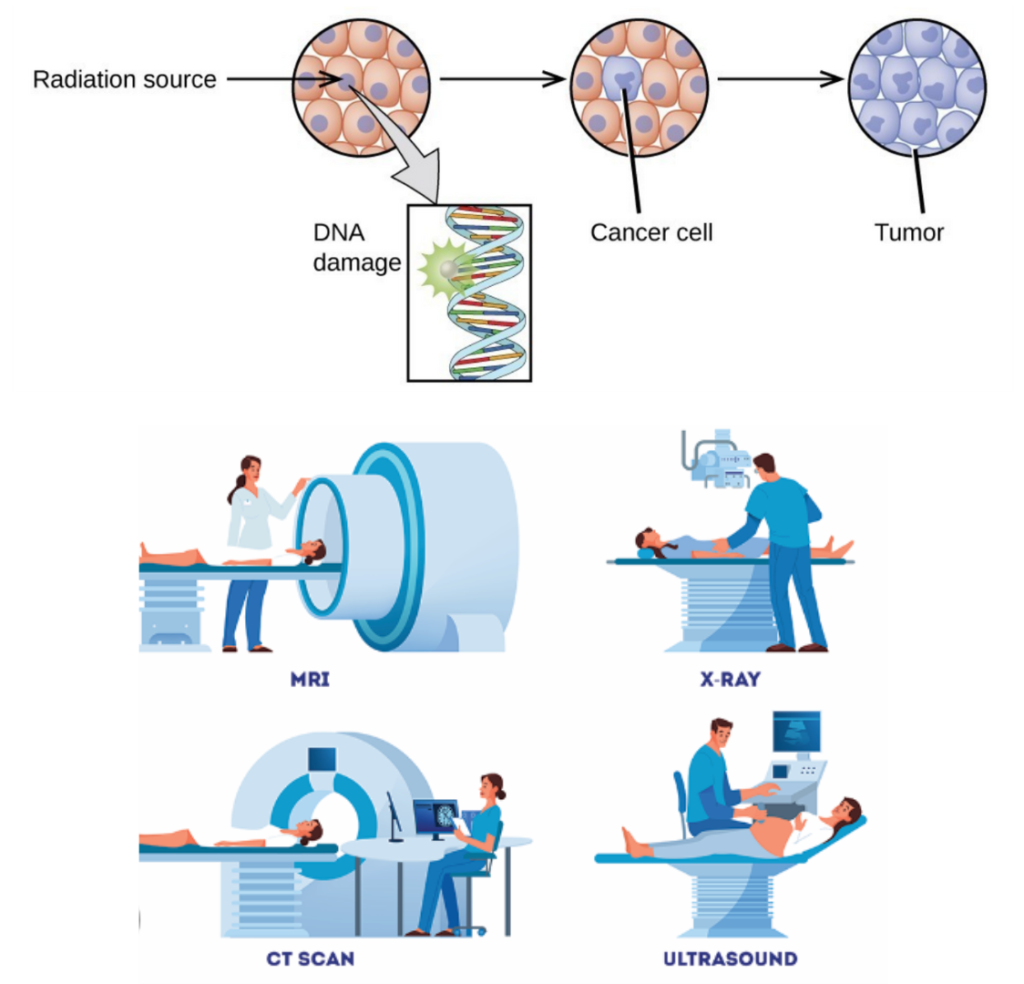
- A large amount of radiation can cause tissue damage like reddened skin and other effects that cannot be seen.
- A small amount of radiation over a long period of time can damage DNA inside a cell, a process known as mutation. Some mutations can cause the cell to malfunction and may cause cancer.
- Not all mutations are harmful, the cell can repair the damage itself if the radiation dose is small.
- In hospitals, radioactivity is used for various purposes. The medical staff working with radiation have limited their exposure in several ways, including increasing their distance from the source and minimising the time they spend in radiation. Staff’s exposure to radiation is closely measured using Dosimeter badges.
- Nuclear Accidents allow the radioactive materials to escape into the environment. Accidents like this are harmful as they can cause people to get irradiated or contaminated.
- Food and water can also get contaminated means that it can enter the food chain. Contamination with radioactive materials of long half-lives poses a greater hazard as the effect will last longer.

Radioactivity in Medicine
- Radioactivity materials are used to diagnose medical issues without having to cut a patient’s body.
- Radioactivity can be used to diagnose cancer using tracers in the body. It can also be used to treat cancer.
Radioactivity tracer detecting medical issue in body, STEPS –
- A radioactive tracer that emits Gamma Rays is introduced into the patient’s body. The tracer is generally injected into the bloodstream. One or more Gamma cameras detect the location of the tracer.
- The tracer finds the location of internal bleeding. The cameras detect the highest gamma radiation, which is where the bleeding occurs.
- Gamma cameras are used to detect tumours, and then the tracer is made using radioactive Glucose molecule.
Frequently Asked Questions
Solution:
When there is spontaneous emission of energy from the unstable nucleus of an atom to become stable, the process is called Radioactivity.
Solution:
Radioactivity is measured using Dosimeter badges.
Solution:
In diagnosis through radioactivity, doctors can find health issues without having to cut the patient’s body. A radioactive substance is introduced into a human’s body, and its radiation is detected/measured through cameras.
Solution:
- Distance: Maintaining a shorter distance from the source.
- Time: The time spent in radiation exposure should be decreased.
- Shielding: Use materials which can block radiation like lead.
Solution:
X-Rays, CT Scans, etc. and other techniques that involve imaging techniques are a source of radiation exposure.
Magnetism and Motor Effect – GCSE Physics
Introduction
- Magnetism is a natural force produced by the movement of electric charges, especially electrons. It creates an invisible region around a magnetic object called a magnetic field, which can attract or repel certain materials, mainly iron, cobalt, and nickel.
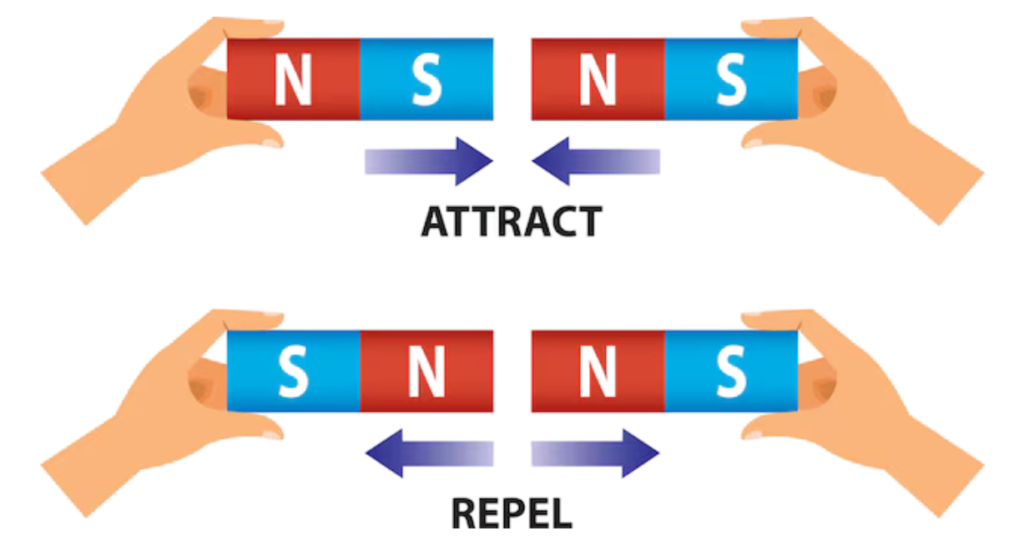
- Every magnet has two ends known as poles — the north pole and the south pole. Like poles repel each other, while opposite poles attract.
- Magnetism plays a key role in many everyday devices, including compasses, speakers, and electric motors. It is also closely linked to electricity, as moving charges can produce magnetic effects.
Magnetism facts and uses of Magnets
Magnetism
- Magnetism is a physical force caused by the motion of electric charges. It creates a magnetic field that can attract or repel certain materials, especially metals like iron. Magnetism is widely used in devices like compasses, motors, and generators.
FACTS ABOUT MAGNETISM
Magnetism comes from moving charges
- When electric charges (like electrons) move, they create a magnetic field around them.
Like poles repel, unlike poles attract
- Two north or two south poles push away from each other, while a north and south pole pull toward each other.
Earth has its own magnetic field
- The Earth behaves like a huge magnet, with a magnetic north and south pole. This is why compasses point north.
Only certain materials are magnetic
- Metals like iron, cobalt, and nickel can be attracted by magnets. Other materials like wood or plastic are not affected.
USES ABOUT MAGNETISM
Electric Motors
- Magnetism is used to create motion in electric motors. When an electric current flows through a coil inside a magnetic field, it experiences a force (motor effect) that makes it spin. This principle is used in fans, washing machines, mixers, and electric vehicles.
Generators
- Generators use magnetism to produce electricity. When a coil of wire moves within a magnetic field, it generates an electric current. This is how electricity is produced in power stations using turbines and magnets.
Magnetic Storage Devices
- Hard drives and some types of memory use magnetism to store data. Information is written and read using tiny magnetic fields that represent binary data (0s and 1s).
What is the working of Motor Effect?
Introduction
- An Electric motor is a device that changes electrical energy into mechanical motion (movement). It works using the motor effect, which is the force that acts on a wire carrying current in a magnetic field.
- Before discussing the principle, let us understand the Rule first.
Fleming’s Left-Hand
- Fleming’s Left-Hand Rule helps us find the direction of motion (force) in an electric motor.
It is used when:
- A current flows through a wire
- The wire is placed in a magnetic field
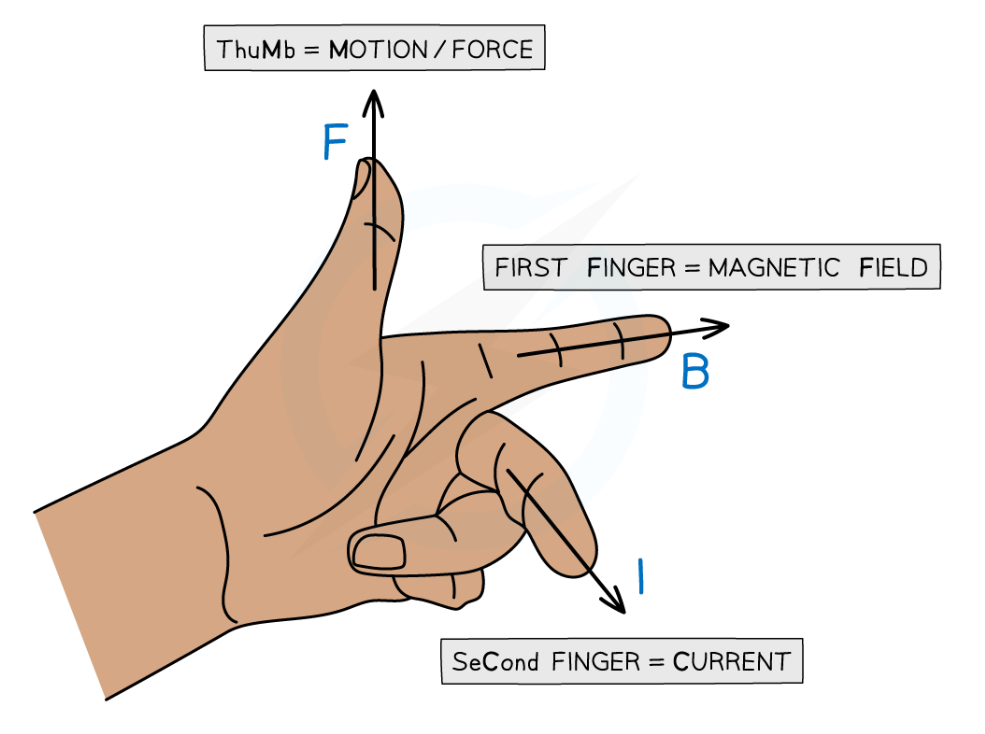
Stretch out your left hand with the:
- Thumb pointing up
- First finger (index) pointing forward
- Second finger (middle) pointing sideways
Example:
If a wire carrying current is placed in a magnetic field, use your left hand like this:
- First finger → magnetic field (from North to South)
- Second finger → direction of current
- Thumb → the direction the wire will move
This rule helps in designing and understanding how electric motors work.
Key Principle:
- Fleming’s Left-Hand Rule helps predict the direction of motion in the motor.
Working:
- An electric motor is a device that converts electrical energy into mechanical energy (motion). It works based on the motor effect, where a current-carrying wire in a magnetic field experiences a force.
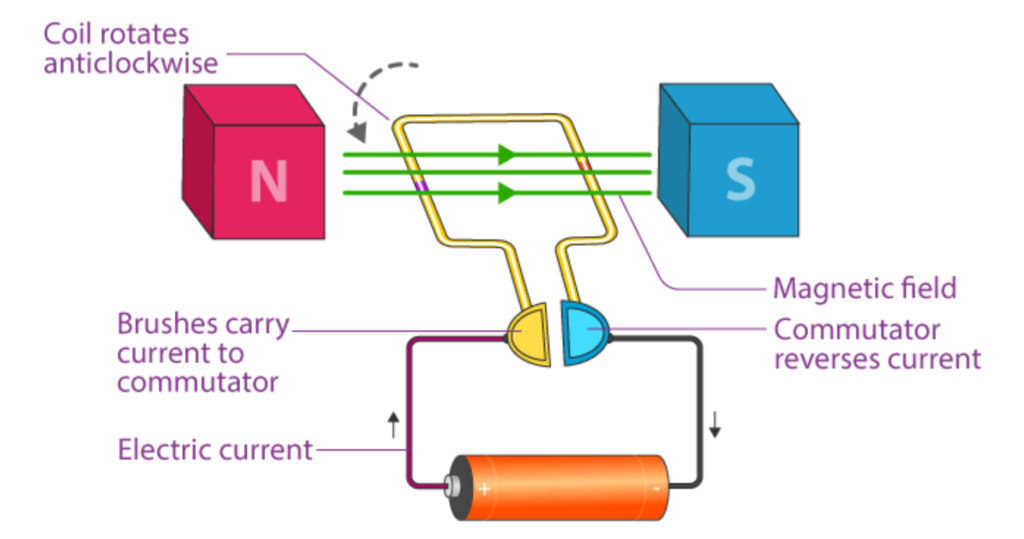
Main Parts of an Electric Motor
Armature (Coil)
- A coil of wire that carries current.
- Placed in the magnetic field and rotates when current flows.
Magnet (Field Magnet)
- Provides a magnetic field (can be permanent or electromagnets).
- Helps create the force that turns the coil.
Commutator
- A split ring that reverses the current direction in the coil every half turn.
- This keeps the armature rotating in the same direction.
Brushes
- Made of carbon, they maintain contact with the rotating commutator and supply current from the battery to the armature.
Battery or Power Supply
- Provides the electric current needed to operate the motor.
Step-by-Step Working of a Motor
- When the motor is powered, current flows through the coil (armature).
- The coil is inside a magnetic field, so the motor effect causes one side to move up and the other side to move down.
- This creates a rotating force, causing the coil to spin.
- The commutator reverses the current in the coil after half a turn, so the rotation continues in the same direction.
- This spinning motion can be used to do mechanical work (like turn a fan or rotate a wheel).
Diagram of Electric Motor
What do you mean by Generator Effect?
Introduction
- The Generator Effect is the process of generating electricity by moving a conductor (like a wire or coil) through a magnetic field. This movement causes an electric current to be induced in the wire.
- Before discussing the principle, let us understand the Rule first.
Faraday’s Law used in a Generator
Faraday’s Law of Electromagnetic Induction states:
- A voltage (or electromotive force) is induced in a coil when it experiences a change in magnetic field. The faster the change, the greater the induced voltage.
Mathematically:

where:
- EMF = induced voltage
- N = number of turns in the coil
- Φ (phi) = magnetic flux
- dΦ/dt = rate of change of magnetic flux.
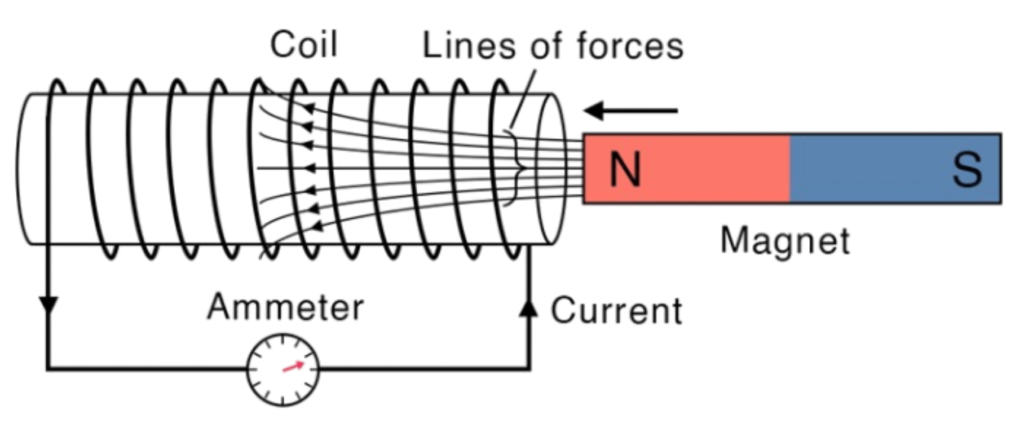
Key Principle:
- The generator effect is based on Faraday’s Law of Electromagnetic Induction, which states:
“Whenever a conductor cuts through a magnetic field or the magnetic field around a conductor changes, a voltage (electromotive force) is induced. If the circuit is complete, current will flow”.
Main Parts of a Generator
Coil (Conductor)
- A loop or winding of wire, usually copper, where current is induced.
Magnet
- Provides the magnetic field. It can be a permanent magnet or an electromagnet.
Motion (Mechanical Energy)
- Either the coil or the magnet is moved to create relative motion between the magnetic field and the coil.
Slip Rings and Brushes (in AC generators)
- Used to transfer the generated current from the rotating coil to the external circuit.
Commutator (in DC generators)
- Used to reverse the current direction every half turn so the output remains in one direction.
Step-by-Step Working of a Generator
Relative Motion:
- The coil is rotated inside a magnetic field, or the magnet is moved near a stationary coil.
Cutting Magnetic Field Lines:
- As the wire moves across the magnetic field, it cuts through the magnetic lines of force.
Induction of Voltage:
- This motion induces a voltage (also called electromotive force) in the wire.
Flow of Current:
- If the coil is part of a closed circuit, the induced voltage causes electric current to flow.
Direction of Current:
- The direction of the induced current depends on the direction of movement and the magnetic field (can be found using Fleming’s Right-Hand Rule).
Diagram of Generator
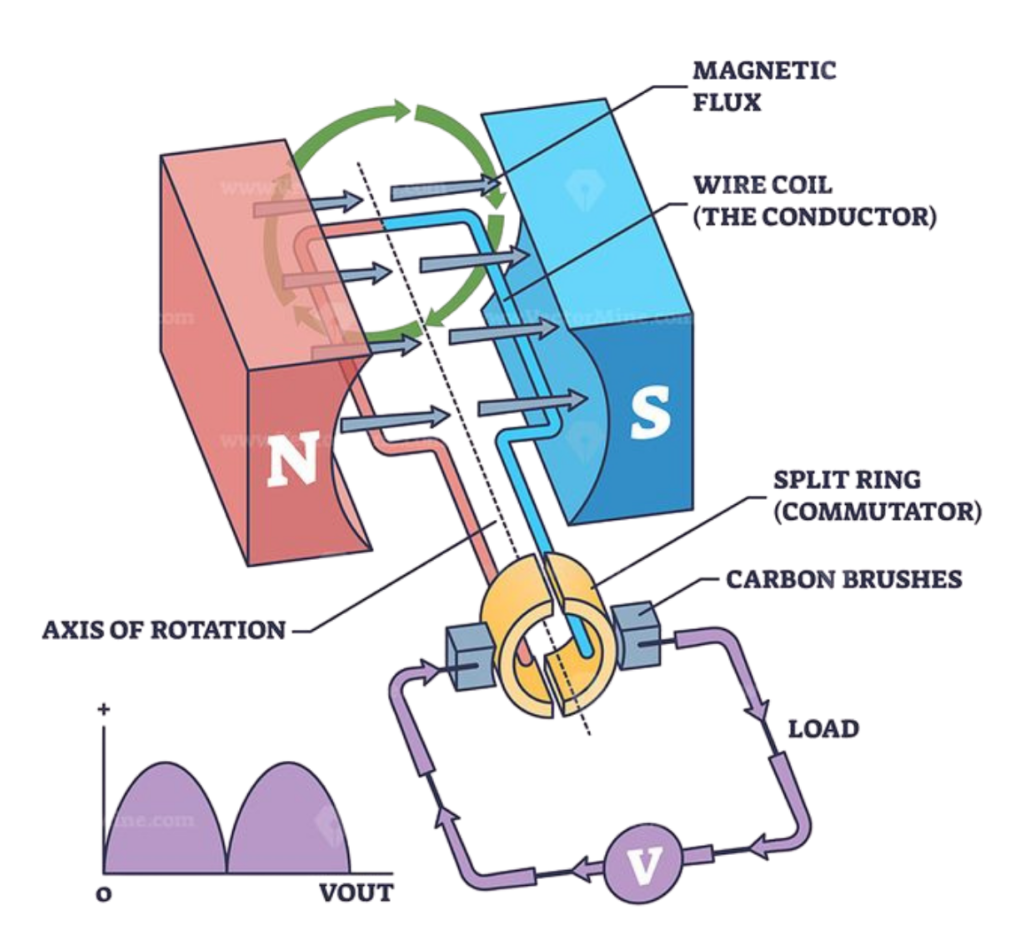
Differentiate between Motor and Generator

Frequently Asked Questions
A magnetic field is the invisible area around a magnet where magnetic forces can be felt. It is shown using field lines going from the north to the south pole.
Fleming’s Left-Hand Rule helps us predict the direction of the force (motion) in an electric motor.
Yes! The Earth has a magnetic field with a north and south pole, which helps guide compasses.
No. You cannot have just one magnetic pole. If you cut a magnet in half, both pieces will still have a north and south pole.
A commutator reverses the direction of current in the coil every half turn so that the motor keeps spinning in one direction.
Fleming’s Right-Hand Rule helps to predict the direction of induced current in a generator.
- Thumb = Motion
- First finger = Magnetic field
- Second finger = Current
Astronomy – GCSE Physics
Introduction to Astronomy
- The Astronomy is branch of Science that deals with the study of Astronomical objects like Stars, Planets and Asteroids etc.
- We can also say that the study of the objects that are beyond Earth’s atmosphere and are present in far away galaxies is known as Astronomy.
- It uses the concepts of Mathematics, Physics and Chemistry in order to explain the origin and the overall evolution of Universe.

What is a Protostar?

- Protostar is a dense core which is very hot and is surrounded by dust and gas.
- Protostar is the early development phase of a star formation.
- The Protostar looks like a Star but its core is not that hot for nuclear fusion to take place.
- As the core of Protostar contracts its temperature rises leading to nuclear fusion and it becomes a main sequence star.
- when protostar rotates it generates a magnetic field.
What is a Star? And Life Cycle of a Star

A glowing ball of hot gas with a high amount of mass is called a Star.
- The most familiar star to us is Sun which give us light and warmth .
Their are many galaxies in the Universe. Our galaxy is Milky way that contains billions of stars just like other galaxies.
Though there are billions of stars in our Galaxy, a few thousands of them are visible to the naked eye from Earth.
Stars shine because there is process that keeps on going on their surface that process is Nuclear Fusion.
Stars seems to twinkle from Earth’s surface because the atmosphere contain many flying dust particles and due to the temperature conditions.

- Life Cycle of a star is process which spans billions of years. The process contains gravitational collapse and nuclear fusion. The process happens in following stages –
Steller Nebulae(Birth): In the beginning there is a large cloud of dust particles and gas which we call as Nebulae. They are measured in light years(Astronomical distance unit). These Nebulae regions collapse due to gravitational forces which forms Protostars. Example: Orion Nebulae

Protostar(Early Development): Protostar is a dense, hot core surrounded by dust and gas. Protostar is the early development phase of a star formation. When the core contracts due to gravitational forces then temperature rises and the process of nuclear fusion can start and then protostar becomes main sequence star.

Main Sequence(Stable Phase): When the environment of protostar is hot and exceeds a range of temperature, then nuclear fusion starts. The hydrogen gets converted into Helium and high energy is released and protostar becomes main sequence, the longest phase of star formation which lasts for billions of years. Here it shines brightly, our sun is currently in this phase.

Red Giant/Super Giant: Huge and massive stars used their fuel fastly, when the fuel is used star starts collapsing within itself.Stars having fuel for further fusion forms Red giant(lesser massive and red coloured) or Super giant(massive and blue coloured).

White Dwarf(medium or low mass stars): When the fuel is exhausted, including Helium, Red Giant becomes unstable and the outer layers move into space, forming planetary nebulae and then the core left behind is called white dwarf which is very dense. The mass of a typical white dwarf is half of the mass of Sun. The White Dwarf cools down over years and becomes Black Dwarf.


Neutron Star/Black Hole: A Neutron Star is the result (remnant) of a big explosion(Supernova) of a massive star. A neutron star has a mass greater than the Sun, which is compressed into a sphere. A Neutron Star is composed of Neutrons (subatomic particles with no electrical charge).

A Black hole is a highly massive object in space with a powerful gravitational pull that even light can not escape it. There’s a boundary around Black Hole(event horizon) which warps spacetime. Anything that crosses that boundary can never come back from Black Hole.
What is Solar System?
- Solar System is a moving system bounded by gravitational forces consisting of sun and planets with their asteroids, comets and dwarf planets in space.
Solar System is magnificent and well moving system.
A large and hot ball is in the center of this system , Sun and its gravitation is so strong that it keeps other planets revolving around it.

The path or the journey that a planet takes around the Sun is called an Orbit.
The Orbit of each planet in the Solar System is elliptical means shaped like an egg.
Each planet of the Solar System do not travel at the same speed around the Sun. The farther is the planet from the sun the more time it takes to complete one orbit (slower speed).

- Mercury is closest planet to Sun and fastest to complete its orbit. It takes 88 Earth days to complete one orbit around the Sun.
Next planet is Venus, the hottest planet in the Solar System takes 224 Earth days to complete the orbit.
Then comes our Earth, planet where life exists takes 365 days to complete one orbit around the Sun.
Mars the 4th planet from sun takes 686 Earth days to complete an Orbit around the Sun.
Jupiter, the biggest planet in the Solar System takes 4332 earth days to complete an orbit.
Saturn, 6th from Sun having glorious ring system takes 10,756 Earth days to complete an orbit.
Uranus, takes 30,687 Earth days and Neptune takes 60,190 Earth days to complete an orbit around the Sun.

What are Dwarf Planets and Asteroid like celestial bodies in Space?
- Our solar system also contains other planets called Dwarf planets, including Pluto, Makemake, Eris, Haumea, and Ceres. They are called Dwarf planets because they are spherical due to their gravitation but are not massive enough to be considered as planets. And just like planets they also revolve around the Sun in their orbits.

Solar System not only contains planets but asteroids, meteoroids, meteors and comets etc. which orbits the sun and contributes to solar system’s complexities. They are-
Comet
Asteroid
Meteoroid
Meteor
Meteorites
- Comets: Comets are big balls of dust, rock and metal. It is surrounded by an icy shell. When comets go too close to something hot like Sun its ice sublimates and hot wind of Sun blows the sublimated ice backwards creating a tail of the Comet.those comet which orbits longer than 200 years are Long Period Comets and Those comet which orbits shorter than 200 years are Short Period Comets. Comets orbit the sun in highly elliptical path.

- Asteroid: Asteroids are just rock, dust and metal. Most Asteroids are formed between the Mars and Jupiter, the region is called as Asteroid Belt. They are dangerous for Earth. They are the reason that caused dinosaurs to go, when an asteroid hit our Earth 65 million years ago.

- Meteoroids: They are rocky floating space remains in Solar System. They are smaller than Asteroids. Some of them come from Asteroid belt whereas some come from the Moon.

- Meteors: We know them as shooting stars. They are just meteoroids that hit the surface of Earth. They are as the size of pebbles. When Meteors hit the surface of Earth they slowly burn and that cause a big flash of light in the sky.

- Meteorites: Meteorites are just Meteoroids that successfully hit the Earth’s surface surviving through its atmosphere. They are dangerous as due to their small size they cannot be detected. Even when their size is is small they travel with high velocity that is why they are dangerous.

Geostationary Satellites
- Geostationary Satellite is placed at an altitude of approximate 36,000 km above the Earth’s surface and just over the equator, the Geostationary satellite moves with Earth (West to East) in an orbit around it. It takes 24 hours to complete one orbit just like Earth take the same amount of time to rotate about its axis once.
- Geostationary satellite is named so because it seems to be stationary when seen from the Earth’s surface while it is moving.

- Applications of Geostationary satellite includes –

Weather Forecasting
Television, Telephone and Radio Broadcasting
Military satellites
Navigation systems, Google Maps etc.
Frequently Asked Questions
Sun( A star in its Main sequence phase, massive and very hot).
They are made from rock and floats in our Solar system, they are of very small size and travel with high velocity. Unlike Meteoroids they may reach the syrface of Earth and hit it causing danger.
The key difference between them is that Comets are surrounded by an icy shell but Asteroids are not.
Shooting Stars is actual are the Meteors falling at the Earth’s
surface but they burn during this journey, hence are not dangerous.
The Asteroid belt is present in between Mars and Jupiter as most of the Asteroids are formed in this region.
Mercury.
Because the Sun is not massive enough to end up as black hole, the Sun needs to be 20 times more massive to become a black hole.
The celestial bodies present in our Universe that are spherical due to their gravity and orbits the Sun are called Dwarf Planets.
A Satellite that is placed over the equator at a distance of 36,000 km from Earth. The satellite moves with Earth in same direction and thus seems to be stationary. There are many applications of these satellites like weather forecasting and google maps etc.
Radioactivity – GCSE Physics
What is Radioactivity?
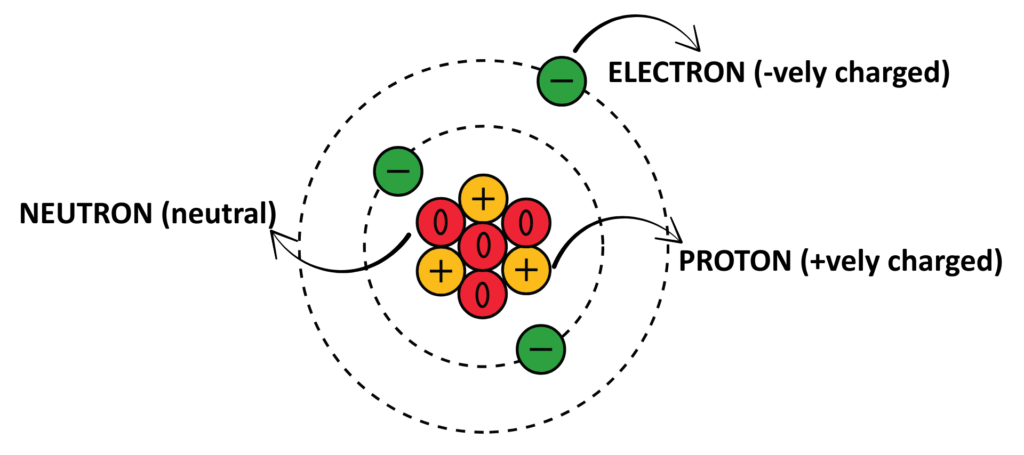
- An Atom is the basic unit of matter. Atom contains Electrons(negatively charged), Protons(positively charged) and Neutrons(neutral) in such a way that the protons and neutrons are present in nucleus and electrons revolve around them in orbitals.
The nucleus is present in center that contributes for mass of an Atom. The nucleus is positively charged due to the presence of Protons.

- When the nucleus of an Atom is Unstable then it releases some of its energy in the form of radiation from nucleus and becomes stable. This process of decay of an Atom is called Radioactivity.

What are Background Radiations?
- The Explosion of low level ionising radiations from the radioactive substances present naturally in our environment and from space.
Sources: The rocks containing small amount of Uranium produces Radioactive gas. The main source is Radon gas. This gas can enter into houses. Most of the high energy rays coming from sun are stopped in the upper atmosphere but some of them reach the surface of Earth and are known as Cosmic rays.


- Variation of Background radiation in different parts of the UK :
- The amount of radioactive gas produced by rocks depends on the rock type.
- Some foods naturally contain radioactive substances.
- In hospitals, X-rays and Gamma-rays are used, which causes background radiation is caused.
- High energy particles coming from the sun when could not be stopped by the atmosphere they reach at the surface of Earth causing background radiations and are called as Cosmic Rays.
How Radioactivity is measured? And Types of Radiation



- Types of Radiation:

There are four kind of Radiations –
1) Alpha Radiation
2) Beta Radiation
3) Gamma Radiation
4) Neutron Radiation
(1) Alpha Radiation (α) – When the nucleus shoots out an Alpha particle (two neutrons and two protons), we know that the helium atom also contains two protons and two neutrons in its nucleus. Thus, the radiation where Helium atom(without any electron) is emitted is called Alpha Radiation.

(2) Beta Radiation(β) –It generally occurs when a neutron in an atomic nucleus turns into proton and electron. The proton remains in the nucleus but the electron escapes the atom.

3) Gamma Radiation (γ) – When an extremely high-energy photon(a small packet of light energy) escapes the nucleus which we call Gamma-ray. It is similar to emission of ordinary light from atom but the gamma ray can damage our surroundings. Gamma-rays involves high energy, The x-rays are similar to gamma-rays but are less energetic.

(4) Neutron Radiation (n) – Here a neutron escapes the nucleus of an atom. This is the most dangerous type of radiation.

# Brief Description about different types of Radiations –
# How can we stop Radiations?

- Alpha radiations consists of heavy and slowly moving particles with a lot of electrical charge, thus they can be easily stopped using a single sheet of paper.
- Beta radiations consist of light and fast moving particles with electrical charge. This means that they can be stopped by thin plates of material.
- Gamma radiations consists of fast moving particles with no electrical charge, they are harder to stop. We use dense material like Lead to stop Gamma-ray.
- Neutron radiations consist heavy particles with no electrical charge. They can travel long distance. We need something that contains large amount of Hydrogen in it.

Applications of Radioactive Substances
Radioactive Substances are dangerous, but they also have many advantages, which is why we can apply them in our real life to resolve some problems, like –
Food naturally contains bacteria that eventually cause them to decompose. Some of them can result in food poisoning, but using gamma-rays we can irradiate them to make it safer to eat.

- They can be used as Tracers. For example, following its path using the GM Tube to detect whether there is leakage from underground pipe. Higher level of radiations are observed at places of leakage.
- Cancer diagnosis by tracers in the body.
- Checking paper thickness.

Frequently Asked Questions
Solution:
- Alpha decay, Beta decay, Gamma decay are primary types of radioactive decay
Solution:
Medical imaging, leakage detection and irradiating food to prevent food poisoning.
Solution:
- No, the ionizing radiation is not always harmful but the higher doses are. Low doses are present naturally in our environment which we call Background radiations.
Solution:
- The main resource is Radon gas from rocks and cosmic rays(sun rays that could not be stopped by Atmosphere).Also the rays used in Hospitals.
Solution:
- Background radiation can enter in a house in multiple ways From the building material, soil and water, cosmic rays, rocks and through cracks etc.
Static Electricity – GCSE Physics
Introduction
- Static electricity happens when electric charges build up on an object’s surface and stay there until they move away.
- It usually occurs when two materials are rubbed together, causing one to gain electrons and the other to lose them.
Real-life Examples:
- Rubbing a balloon on your hair makes your hair stand up, showing static electricity.

- When we comb our dry hair, the comb can attract small bits of paper.

What is Static Electricity?
- Static electricity refers to electric charge that does not move and remains stored on the surface of insulating materials until it can discharge. It happens when two materials rub against each other,
- causing electrons to jump from one object to another.
How Does It Work?
- We know that each matter is made of atoms, which contain protons (+) and electrons (–). When two different materials rub together,
- electrons can transfer, leaving one object positively charged and the other negatively charged.

- Opposite charges attract, so the charged objects may stick together or create a tiny spark when they get close to something with the opposite charge.
Key points:
Static electricity involves charges at rest (unlike current electricity where charges flow).
The charges can cause attraction or repulsion between objects.
It can lead to discharge (sparks or shocks) if the built-up charge finds a path to move.
What is Charging by Induction?
- Charging by induction is when an object gets charged without touching it.
When a charged object is brought near a neutral object, the charges inside the neutral object rearrange, with opposite charges moving closer and like charges moving away.
So that one side of the object becomes charged without making any direct contact.
Example:
- When a negatively charged balloon is brought near the wall, the electrons in the wall move away because like charges repel.
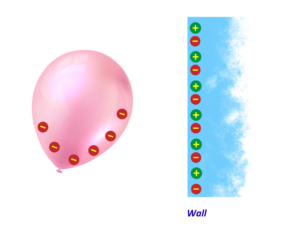
- This leaves the surface of the wall positively charged, which attracts the negatively charged balloon and makes it stick to the wall.
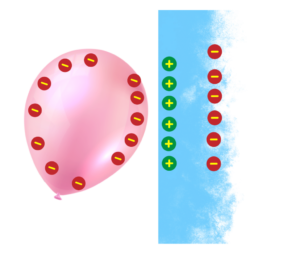
What are Electric Fields?
- An electric field is an invisible area around a charged object
- where it can exert a force on other charges. It shows how a charged object can attract or repel other charged objects without touching them.
- The strength of the electric field is stronger closer to the charged object and gets weaker as you move away.
Representation of Electric field lines:
- The lines point away from positive charges and toward negative charges.
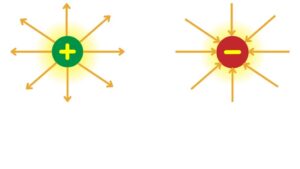
- The closer the lines, the stronger the field.
- The direction of the lines shows the direction a positive charge would move in the field.
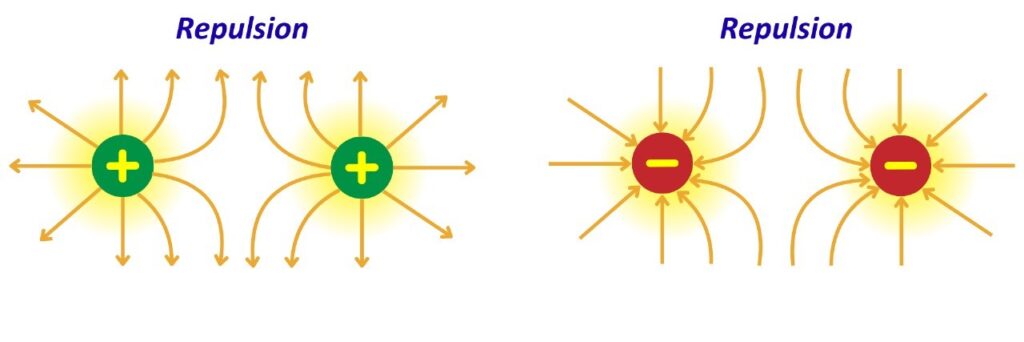
- Like charges repel each other in an electric field, pushing away from each other.
Why Do Sparks, Shocks, and Lightning Occur?
- Sparks, shocks, and lightning happen when a large amount of static charge suddenly moves to balance out charges.
- When a charged object has a high potential difference compared to its surroundings, the electric field becomes strong enough to make electrons jump through the air, which is normally an insulator.
Sparks
- Sparks happen when charges jump through the air, like when you see a small spark while touching a metal door handle.

Shocks
- Shocks are what we feel when the charge moves through our body to the ground.

- A huge discharge of static electricity from clouds to the ground balancing the charge difference.

Dangers and Uses of Static Electricity
Dangers
Sparks can cause fires or explosions near petrol or gas.
Can give small electric shocks.
Can damage electronics if charges jump suddenly.
Uses:
Photocopiers and printers use static electricity to stick ink to paper.

- Air purifiers use static to catch dust.

- Used in defibrillators to restart the heart.
Frequently Asked Questions
Solution:
- Static electricity is when electric charges build up on the surface of an object and stay there until they move away.
Solution:
Objects get charged when they are rubbed together, transferring electrons from one to the other.
Solution:
- It is when a charged object causes charges in another object to rearrange without touching it, making one side charged.
Solution:
- An electric field is the area around a charged object where it can push or pull other charges.
Solution:
- Because the electric field pushes like charges apart and pulls opposite charges together.
Solution:
- They happen when built-up static charge suddenly moves through the air to balance charges, causing a spark or shock.
Solution:
- Useful in photocopiers, spray painting, and air purifiers. Dangerous because it can cause shocks, fires, or damage electronics.
Electrical Safety – GCSE Physics
Introduction
- Electricity is a powerful form of energy used in our daily lives for lighting, heating, and operating devices.
- Electrical safety means being careful around electricity to avoid accidents, injuries, or even death.
- It can be dangerous if not used properly, which is why it is important to learn how to handle electricity safely.
What is an Electrical Safety?
- Electrical safety means taking precautions to prevent electric shocks, burns, fires, and damage to appliances when using electricity.
- The earth wire is used for safety because it carries excess current to the ground and stops the metal parts of appliances from giving electric shocks.
- Electricity can travel through the human body, causing serious injuries or death if safety is ignored.
- Learning electrical safety helps to avoid accidents, save lives, and protect property.
Where is Electrical Safety Important?
- Homes (using plugs, switches, and appliances safely).
- Schools (science labs, computer labs).
- Workplaces (offices, factories, hospitals).
- Construction sites (using heavy electrical equipment).
Electrical Safety Precautions:
- Turn off the switch before plugging in or removing a plug.
- Do not overload sockets with too many devices.
- Do not insert objects into plug sockets.

What is an Electrical Plug?
- An electrical plug is a safely device used to connect electrical appliances to the mains electricity supply through a socket.
- It allows the appliance to receive electricity safely from the socket.
- You can easily unplug it whenever you want to stop using your appliance, whether it’s a –

What is it made of, and why is it used?
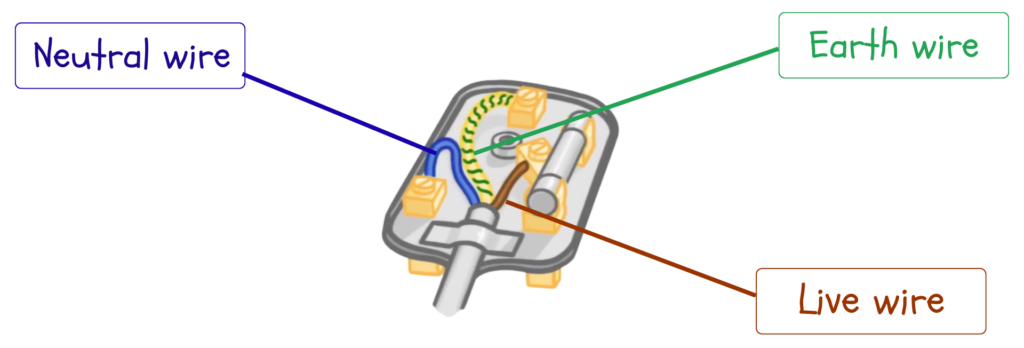
- A plug is made of plastic or rubber for outer casing as insulator to prevent electric shocks. It contains Brass or copper pins to conduct electricity efficiently.

- Inside a plug, three wires are connected in which each wire is made of copper and covered with colored insulation to prevent shocks and short circuits.
- It is used to safely connect appliances to the power supply and allows easy disconnection when needed.
Wires Used in a Plug and Their Features
- Wires are flexible material used as conductors to carry electric current safely to and from devices.
- They are coated with colored plastic to identify their function easily and for safety.
The Three Essential Wires:
1. Live Wire (Brown / Red)
- Carries current from the power supply to the appliance. It is at high voltage.
- Most dangerous wire as it is always live when the switch is on.

2. Neutral Wire (Blue / Black)
- Completes the circuit by carrying current back to the supply.
- It is at or near zero voltage. Helps to complete the flow of electricity safely.

3. Earth Wire (Green/Yellow)
- Safety wire connected to the metal casing of the appliance.
- It carries current to the ground if there is a fault.
- Prevents electric shocks by ensuring the appliance’s body does not become live.

Role of the Earth Wire in Electrical Safety
- The earth wire (or ground wire) is an important part of electrical safety.
- It gives a safe path for extra or faulty current to go into the ground.
- It is useful during short circuits, damaged wires, or insulation failure.
- Without it, current may pass through metal parts of appliances and give shocks.
- The earth wire prevents shocks by safely sending the current into the ground.
- It helps protect people from electric shocks and accidents.
When an earth wire is properly installed, it prevents us from happening:
- If a live wire accidentally comes in contact with the metal body of an electrical appliance, the earth wire immediately carries the fault current down into the ground.
- This prevents electric shocks, as the metal casing does not become live.
- It also activates the fuse or circuit breaker, which cuts off the electricity supply to prevent further danger.
Real-life Example:
- If an iron doesn’t have an earth wire and has a fault, the current can pass through your body, causing a dangerous shock. But if it has an earth wire, the current safely goes into the ground. The fuse will then switch off the power, keeping you safe.

What are Direct Current and Alternating Current?
- Electricity flows in two primary forms: Direct Current (DC) and Alternating Current (AC).
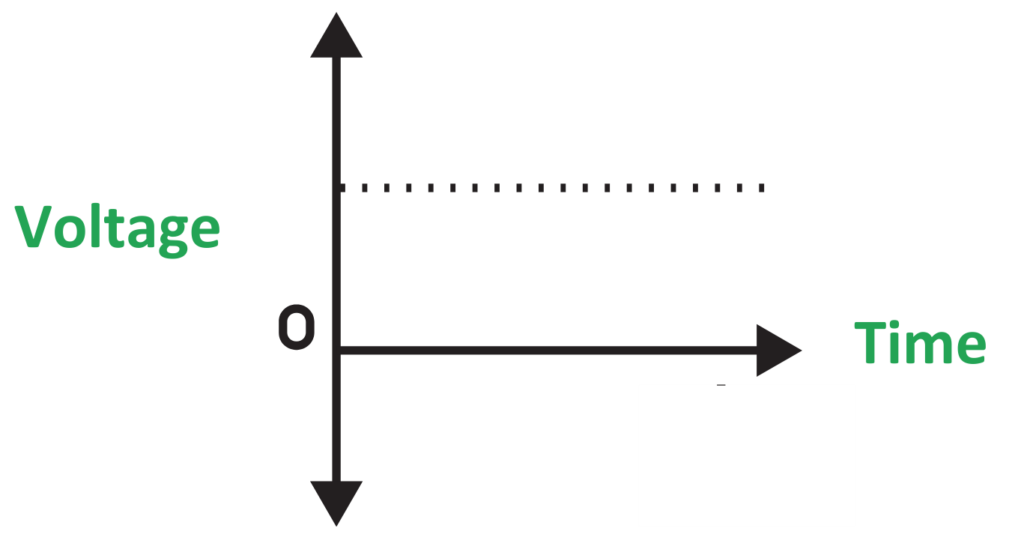
Direct current:
- Definition: Electricity where current flows in one direction only.
- Sources: Batteries, solar cells, DC generators, and electronic devices.
- Applications: Used in torches, remote controls, and phones (via batteries).
Alternating current:
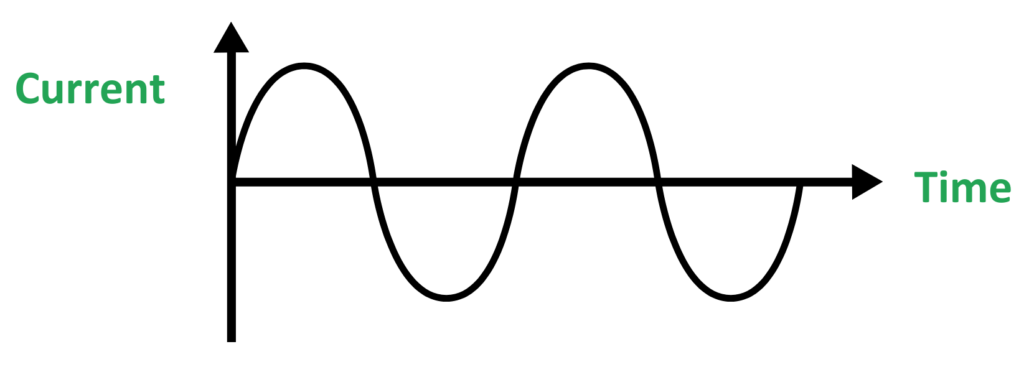
- Definition: Electricity where current changes direction continuously (back and forth).
- Sources: Power plants, generators, household outlets.
- Applications: Used in homes, schools, and industries.
What are Fuses and How do they Ensure Safety?
- A fuse is a protective device made of a thin wire that melts when too much current flows through it, breaking the circuit.
- It protects appliances and users from damage caused by excessive current.
How Does a Fuse Work?
- A fuse is connected in series with the live wire so that all the current flows through the fuse before reaching the appliance.

- The fuse wire has a low melting point, so it will melt if too much current flows, breaking the circuit and stopping electricity.
Example with a Microwave:
- Suppose we are using a microwave that normally uses 10 A current.
- A 13 A fuse is placed in the plug to protect it, as it allows normal current (10 A) to pass safely.

- If a fault occurs and the current increases to 15 A, the fuse wire will heat up and melt.

- This breaks the circuit, stopping the flow of electricity, preventing overheating or fire in the microwave.
- Then we can replace the fuse with a new 13 A fuse after fixing the fault to use the microwave again safely.
What are Circuit Breakers and Their Role in Safety?
- A circuit breaker is an automatic protective device that cuts off the electricity when the current in a circuit becomes too large.
- Circuit breakers do not need to be replaced after they trip; they can be reset and used again.
How Does a Circuit Breaker Work?
- A circuit breaker also connected in series with the live wire of a circuit.

- Inside it, an electromagnet strip detects when the current exceeds the safe limit.
- When the current becomes too high, the electromagnet pulls a switch or the strip bends, opening the circuit and stopping the current.
- This prevents overheating, fires, and damage to appliances.
Example with a Washing Machine:
- A washing machine using 8 A normally is protected by a 10 A circuit breaker.
- If the current rises to 15 A due to a fault, the breaker trips to stop electricity and can be reset after fixing the fault.
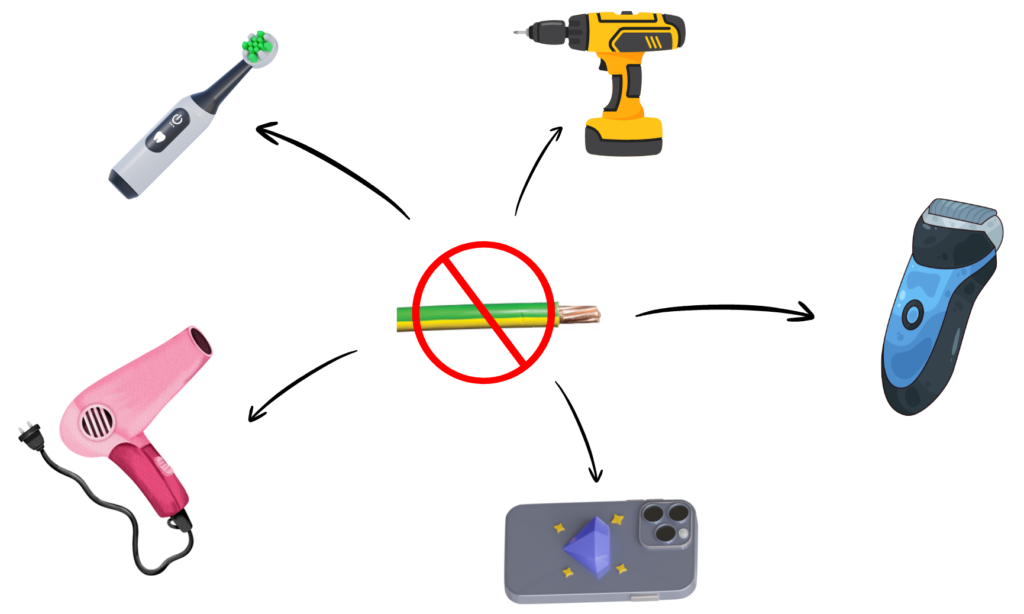
What is Double Insulation in Electrical Devices?
- Double insulation means an electrical device has two protective coverings, so we don’t get an electric shock while using it.
- It is used in appliances that do not need an earth wire for safety.
How Does Double Insulation Work?
- The first layer covers the wires inside the appliance to keep electricity inside the wires.
- The second layer is the plastic outer body of the appliance.
- Even if the first layer breaks, the outer layer keeps you safe from touching electricity.
Example:
- Devices which do not have an earth wire but remain safe to use due to double layer of protection are:
Frequently Asked Questions
Solution:
To prevent electric shocks, burns, and fires while using electricity.
Solution:
Water conducts electricity, increasing the risk of shock.
Solution:
It prevents electric shocks by carrying excess current safely to the ground during a fault.
Solution:
A device used to connect appliances to the mains supply safely.
Solution:
Some plugs have a fuse to break the circuit if too much current flows.
Solution:
For safety and to identify their purpose (live, neutral, earth).
Solution:
Yes, if handled unsafely, it can cause electric shock and burns.
Solution:
Yes, if handled unsafely, it can cause electric shock and burns.
Solution:
Electric current that flows in one direction only.
Table of Content
- Introduction
- What is an Electrical Safety?
- What is an Electrical Plug?
- Wires Used in a Plug and Their Features
- Role of the Earth Wire in Electrical Safety
- What are Direct Current and Alternating Current?
- What are Fuses and How do they Ensure Safety?
- What are Circuit Breakers and Their Role in Safety?
- What is Double Insulation in Electrical Devices?
Atomic Structure – GCSE Physics
Introduction
- An Atom is the fundamental building block of all matter. It generally represents the smallest unit of an element that retains its chemical properties.
- Despite their incredibly small size, atoms form the foundation of all substances in the universe, from the air we breathe to the stars in the sky.
Structure Of An Atom
- The Structure of an atom describes how its fundamental particles—protons, neutrons, and electrons—are arranged, forming the basis of all matter in the universe.
Related terminology
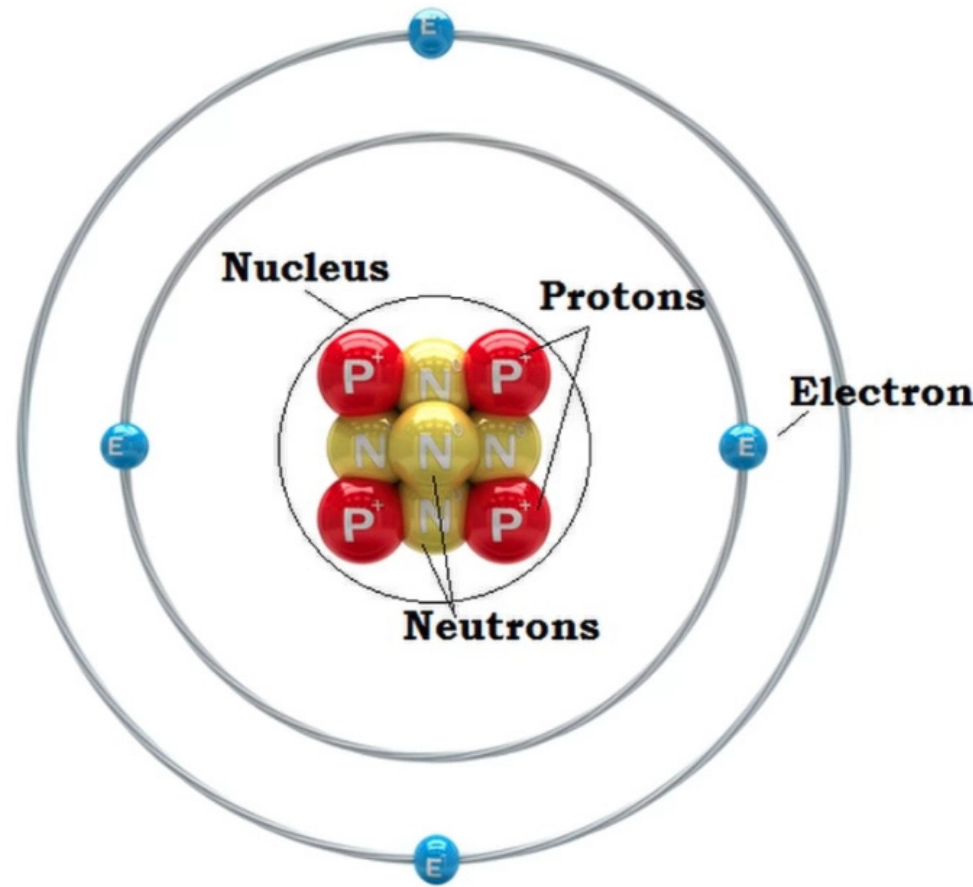
1. Nucleus:
- Located at the center of the atom; contains most of the atom’s mass.
2. Protons:
- Positively charged particles found in the nucleus.
- The number of protons = atomic number.
3. Neutrons:
- Neutral particles also located in the nucleus.
- Protons + Neutrons = Mass number.
4. Electrons:
- Negatively charged particles that orbit the nucleus in energy levels or shells.
5. Electrically Neutral:
- In a stable atom, the number of electrons equals the number of protons.
6. Atomic Stability:
- The arrangement of electrons determines the atom’s chemical reactivity and bonding behavior.
Atomic Particles Of An Atom
- There are three subatomic particles of an atom that are:
- Protons
- Neutrons
- Electrons
- These three subatomic particles work together to define the structure, properties, and behavior of every atom.
Let us understand about them in a more detailed way.
Protons:
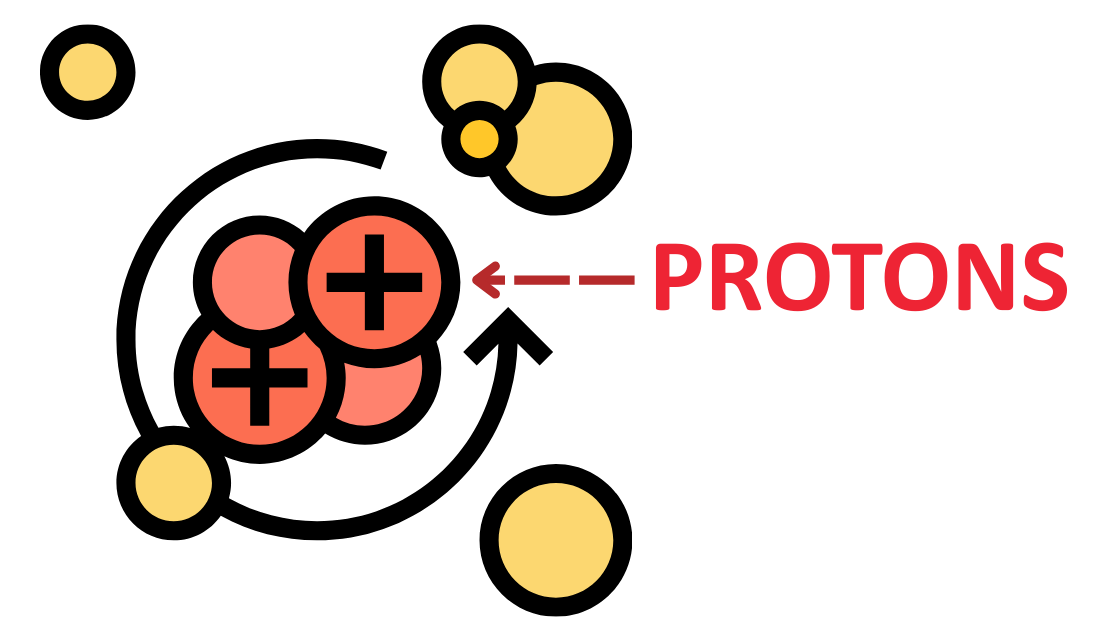
Definition:
- Protons are positively charged subatomic particles found in the nucleus of an atom.
Charge:
- Each proton carries a +1 elementary charge (+1e).
Mass:
- Approximately 1 atomic mass unit (1 amu) or 1.67 × 10-27 kg, slightly less than a neutron.
Role in Atomic Identity:
- The number of protons in an atom defines the atomic number and thus the type of element.
- (e.g., 1 proton = hydrogen, 6 protons = carbon)
Neutrons
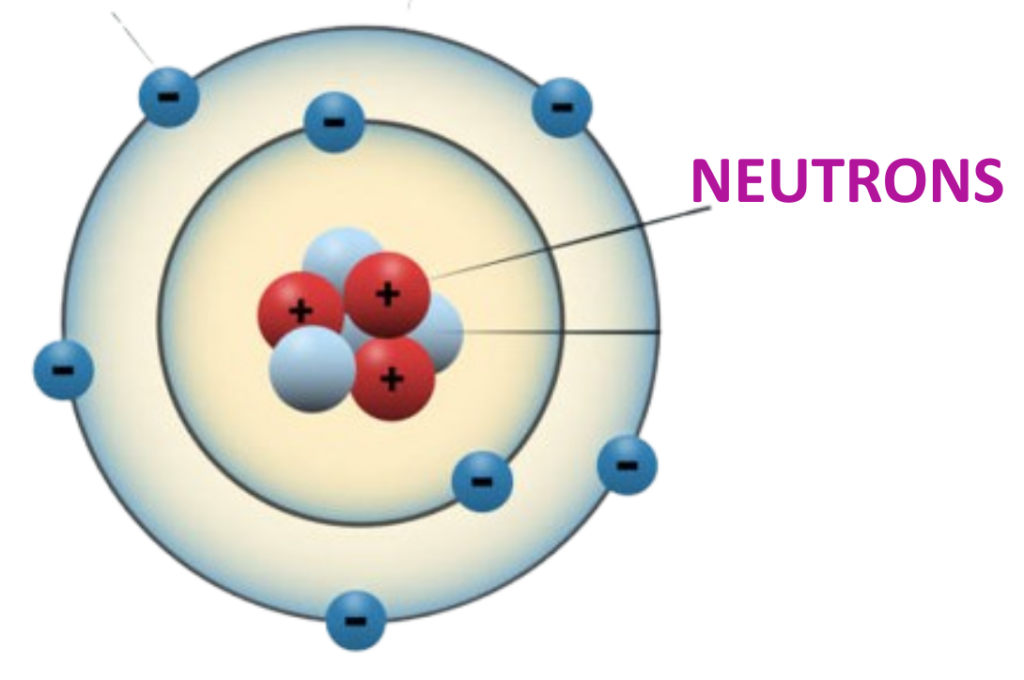
Definition:
- Neutrons are neutral subatomic particles found in the nucleus of an atom.
Charge:
- Neutrons carry no charge (they are electrically neutral).
Mass:
- Neutrons have a mass of approximately 1 atomic mass unit (1 amu).
Role in Atomic Stability:
- Neutrons contribute to the mass of the atom and play a key role in maintaining the stability of the nucleus by reducing the repulsion between positively charged protons.
Isotopes:
- Neutrons help define the isotope of an element. Atoms of the same element with different numbers of neutrons are called isotopes.
- (e.g., Carbon-12 and Carbon-14 are isotopes of Carbon.)
Electrons
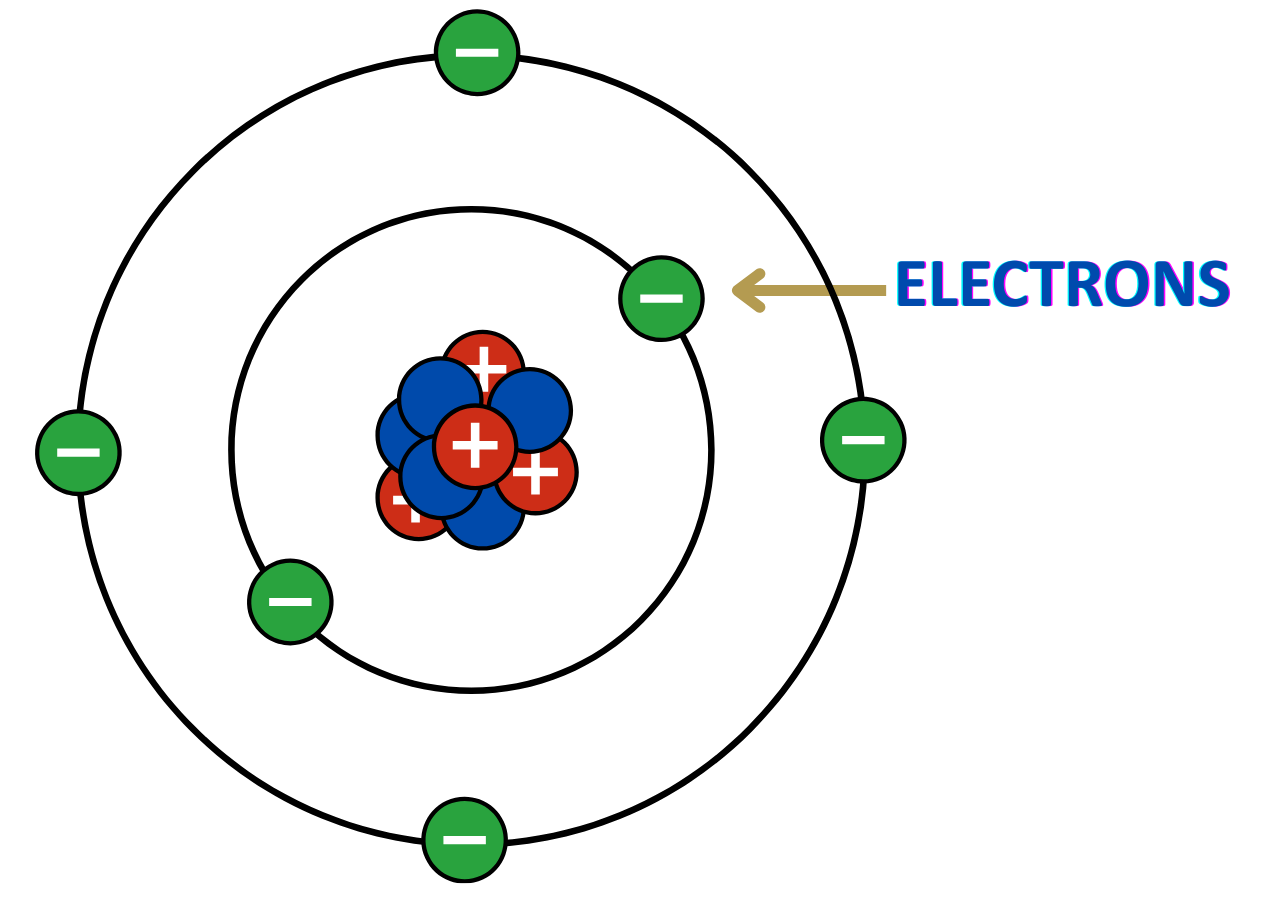
Definition:
- Electrons are tiny, negatively charged subatomic particles found in atoms.
Charge:
- Each electron carries a -1 elementary charge.
Mass:
- Electrons have a very small mass (9.11 × 10⁻³¹ kg), about 1/1836 the mass of a proton.
Location in Atom:
- They orbit the nucleus of an atom in regions called electron shells or energy levels.
Role in Atoms:
- Electrons determine how atoms interact chemically — they are involved in bonding and reactions.
Electricity and Current:
- Moving electrons in a conductor create electric current.
Examples of Structures of Some Elements
- Here’s some examples of elements along with their atomic structures.
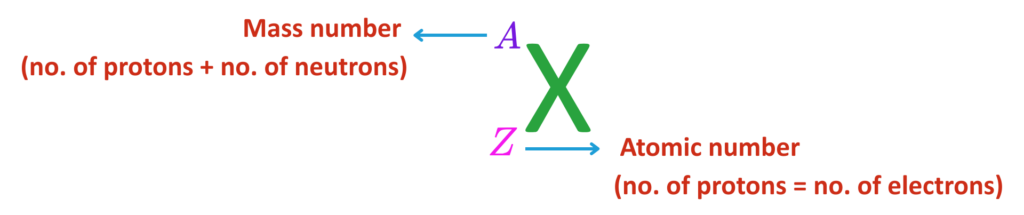
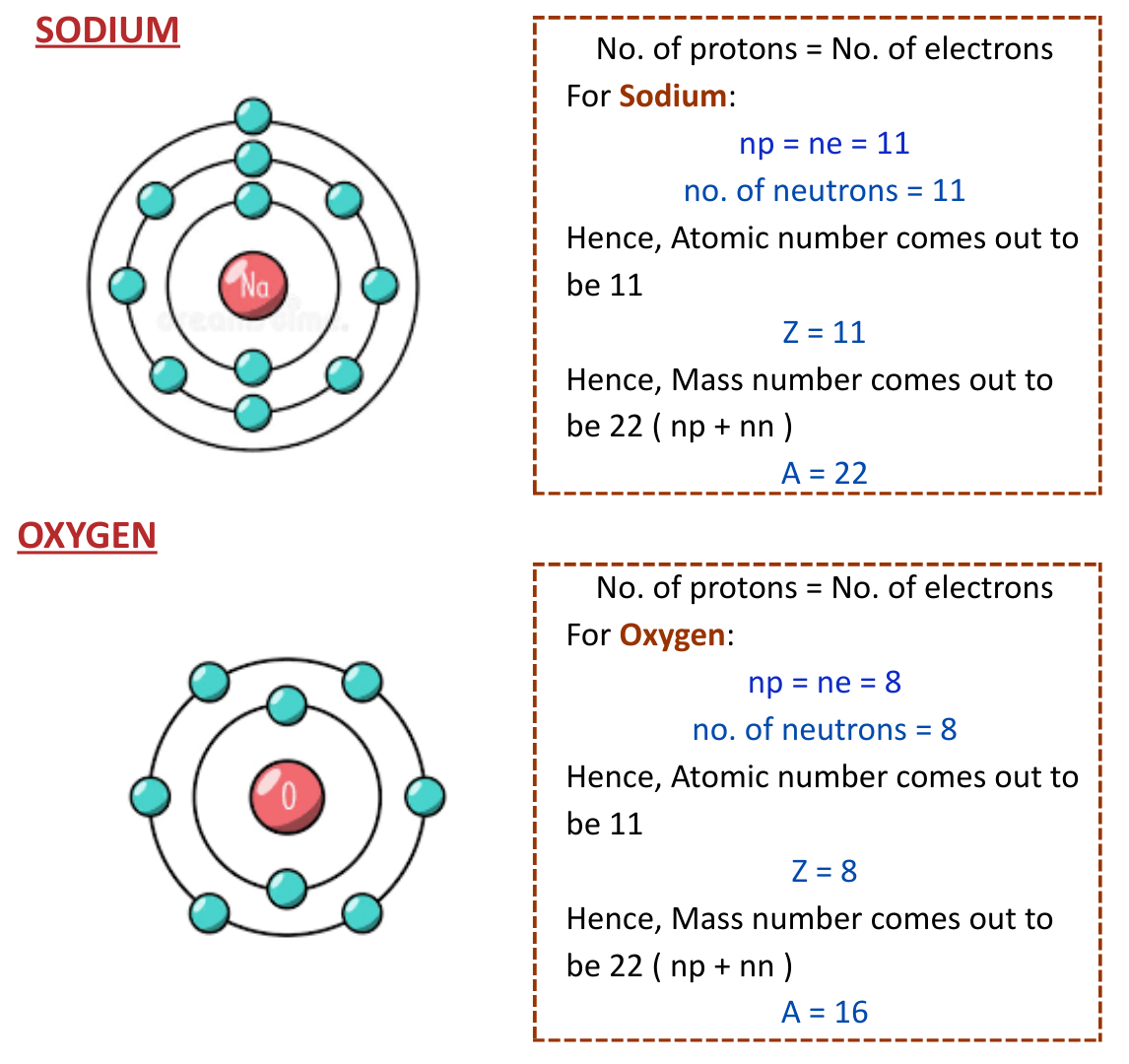
Isotopes
- Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. Basically, isotopes contains same atomic number but they differ in mass number due to uneven number of neutrons.
Key Points:
- Same Element: Same number of protons (same atomic number)
- Different Mass: Due to different numbers of neutrons
- Examples:
- Carbon-12, Carbon-13, and Carbon-14 are all isotopes of carbon.
- All have 6 protons
- Carbon-12 has 6 neutrons, Carbon-13 has 7, Carbon-14 has 8
- It can be explained diagrammatically as:
- Carbon-12, Carbon-13, and Carbon-14 are all isotopes of carbon.
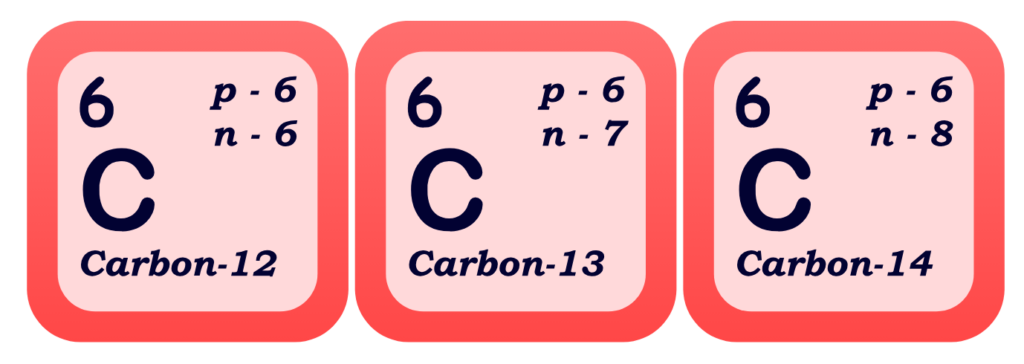
- Some isotopes are stable, while others are radioactive (they decay over time, releasing radiation)
Uses of Isotopes:
- Medical: Radioactive isotopes in cancer treatment and diagnostic imaging (e.g., iodine-131)
- Archaeology: Carbon-14 dating of ancient artifacts
Ionisation Energy
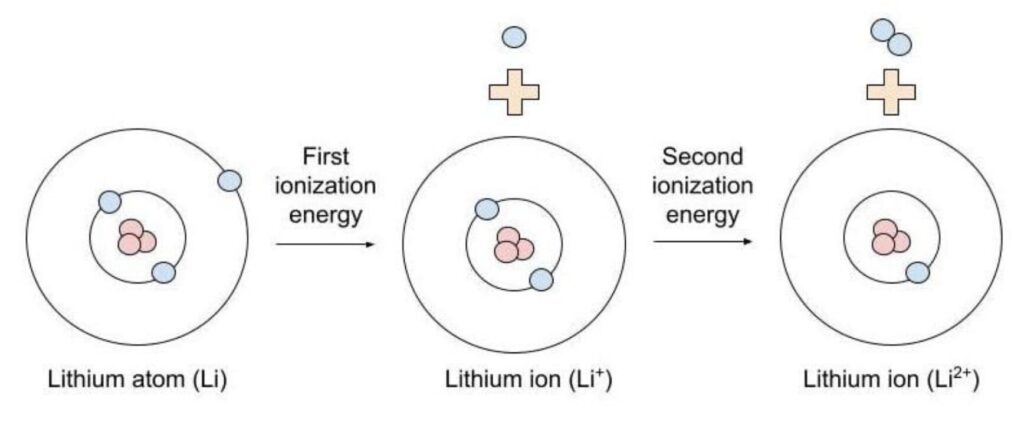
- Ionisation energy (also spelled ionization energy) is the amount of energy required to remove an electron from a gaseous atom or ion in its ground state.
- It is a measure of how strongly an atom holds onto its electrons.
Key Concepts:
- First Ionisation energy: Energy needed to remove the first electron from a neutral atom.
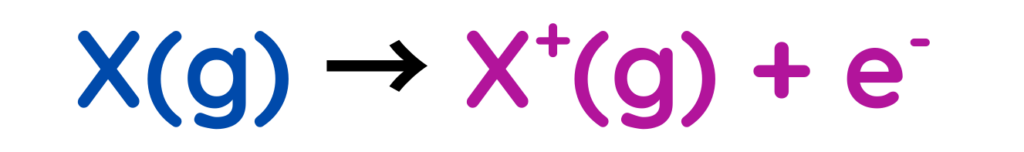
- Second Ionisation energy: Energy required to remove a second electron from a positive ion.
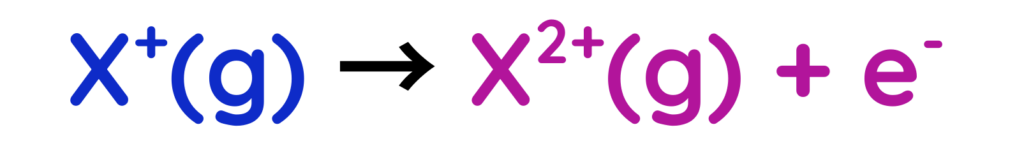
Factors Affecting Ionisation Energy:
- Nuclear charge (more protons = higher ionisation energy)
- Atomic radius (larger radius = lower ionisation energy)
- Electron shielding (more inner shells = lower ionisation energy)
- Electron configuration (full or half-full subshells can affect stability)
Frequently Asked Questions
Solution:
An atom is the smallest unit of matter that retains the properties of an element. It consists of a nucleus (protons and neutrons) and electrons in orbitals around the nucleus.
Solution:
- Proton: Positive charge (+1), found in the nucleus
- Neutron: No charge (neutral), found in the nucleus
- Electron: Negative charge (–1), found in orbitals around the nucleus.
Solution:
The atomic number (Z) is the number of protons in an atom. It determines the element.
Solution:
The mass number (A) is the total number of protons and neutrons in an atom.
Solution:
Isotopes are atoms of the same element (same number of protons) but with different numbers of neutrons (different mass numbers).
Solution:
Ionisation energy is the energy required to remove an electron from a gaseous atom or ion.
Solution:
Electrons occupy specific energy levels around the nucleus. Each level can hold a certain number of electrons (e.g., 2 in the first, 8 in the second).
Electromagnetic Induction – GCSE Physics
Introduction
- Electromagnetic induction is the process of generating an electric current by changing the magnetic field around a conductor.
- This fundamental concept links electricity and magnetism and was first discovered by Michael Faraday in 1831.
What Happens in Electromagnetic Induction?
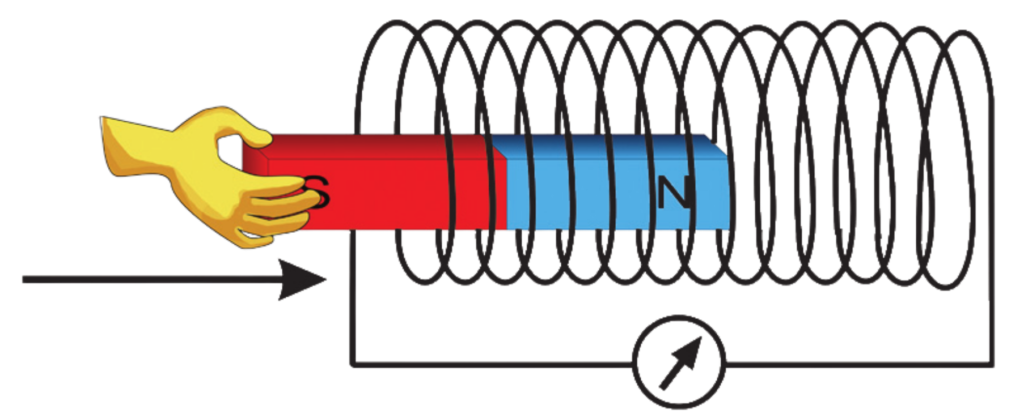
- When a wire or coil is placed in a changing magnetic field, or if a magnet moves near the coil, it causes electrons in the wire to move. This movement of electrons creates an electric current. This is known as induced current, and the voltage produced is called “Induced Electromotive Force (EMF)”.
How Induction Occurs in Electromagnets?
Electromagnet
- An Electromagnet is a type of magnet created when electricity flows through a wire.
- It works only when the current is ON, making it a temporary magnet.
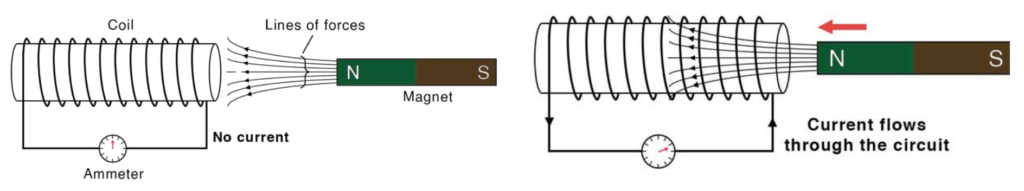
How Induction occurs:
- Induction in electromagnets happens when a changing magnetic field—created by the electromagnet—induces an electric current in a nearby conductor.
Step by Step Explanation:
- Step #1: Electromagnet creates a magnetic field: When electric current flows through a coil of wire (usually wrapped around an iron core), it becomes an electromagnet and produces a magnetic field.
- Step #2: Change in magnetic field: If the current in the electromagnet changes, the magnetic field around it also changes.
- Step #3: Nearby conductor affected: If there is a wire or coil near this changing magnetic field, the magnetic lines of force cut through the conductor.
- Step #4: Induced current: This change causes electrons in the nearby conductor to move, creating an induced current. This is electromagnetic induction.
Key Facts About Electromagnetic Induction
Key Facts about Electromagnetic Induction
- Discovery: Introduced by Michael Faraday, who demonstrated that moving a magnet near a coil induces an electric current.
- Faraday’s Law: The induced voltage is directly proportional to the rate of change of magnetic flux.
- AC-Friendly: Alternating current (AC) is ideal for induction due to its continuous change in direction.
- Contactless Power: Induction enables power transfer without direct electrical connections.
- Efficiency Factors: Induced current increases with faster motion, stronger magnets, or more coil turns.
- Temporary Effect: The induction only occurs as long as the magnetic field is changing.
Uses of Electromagnetic Induction
- Used in electric generators to produce electricity
- Powers transformers to change voltage levels
- Drives induction motors in fans, pumps, and machines
- Enables wireless charging in smartphones and devices.
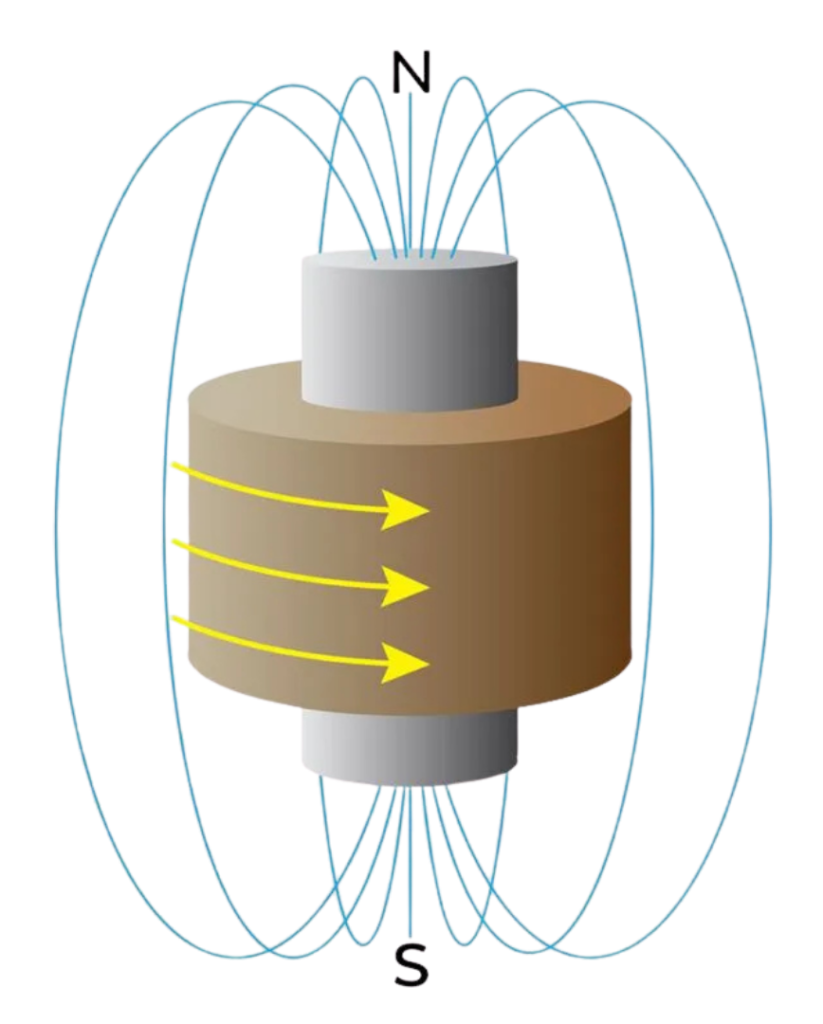
What Do You Mean By EM Coil And EM Currents?
EM Coil
- An EM coil is a wire wound in loops or spirals, often around an iron core, used to create or respond to magnetic fields.
- When current flows through the coil, it acts like a magnet (called an electromagnet).
- When the magnetic field around the coil changes, it can also generate current by induction.
- EM coils are essential in inductors, motors, speakers, and wireless chargers.
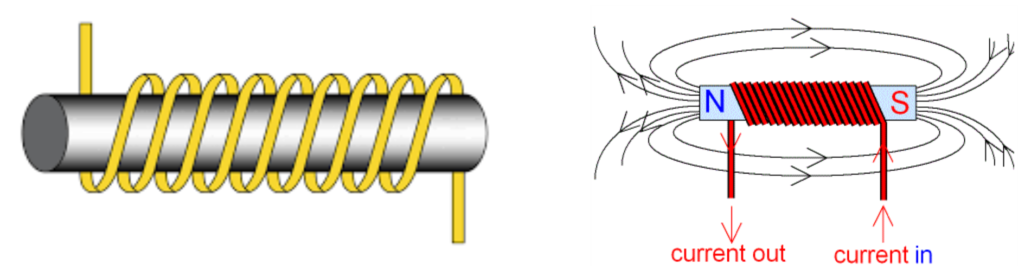
EM Current
- EM currents refer to electric currents produced due to electromagnetic induction. These are not regular currents from a battery, but are induced when a magnetic field around a conductor changes.
- When a magnet moves near a coil, or the coil moves in a magnetic field, electrons in the wire start moving.
- This motion creates an induced current, called an electromagnetic (EM) current.
- EM currents are used in generators, transformers, and induction motors.
Faraday’s Law of Induction
Introduction:
- Faraday’s Law states that:
“An electric current is induced in a conductor whenever it experiences a change in the magnetic field around it”.
Faraday’s Law of Induction
Faraday’s Law of Electromagnetic Induction states:
- A voltage (or electromotive force) is induced in a coil when it experiences a change in magnetic field. The faster the change, the greater the induced voltage.
Mathematically:

where:
- EMF = induced voltage
- N = number of turns in the coil
- Φ (phi) = magnetic flux
- dΦ/dt = rate of change of magnetic flux.
What Does Faraday’s Law Say?
Faraday’s First Law:
- Whenever a magnetic field passing through a coil or conductor changes, an electromotive force (EMF) is induced in the conductor. If the circuit is closed, this EMF causes an electric current to flow.
Faraday’s Second Law:
- The magnitude of the induced EMF is directly proportional to the rate of change of magnetic flux through the coil.
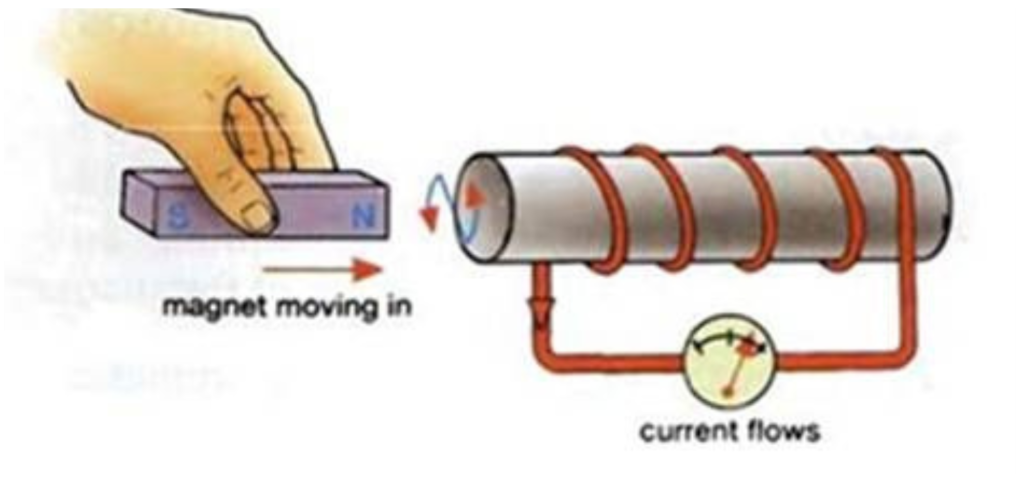
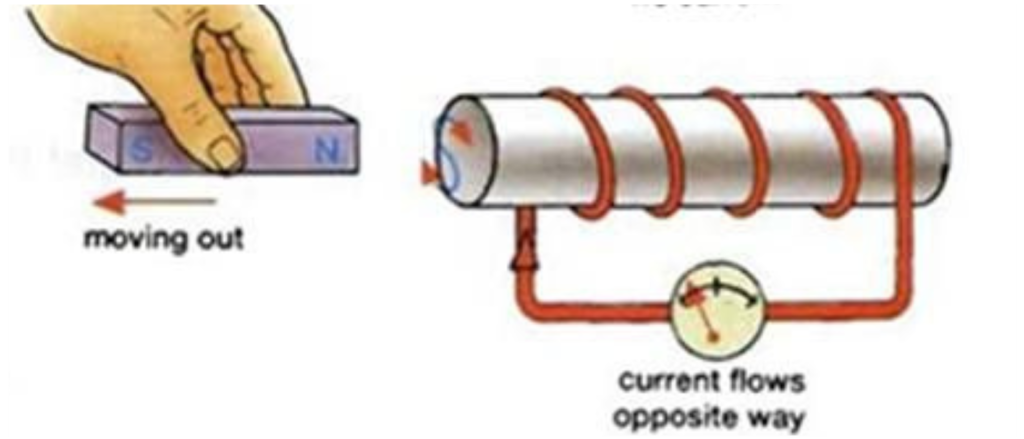
There must be 3 Scenarios that can happen in an Electromagnet
- Magnet moving in
- Current flows
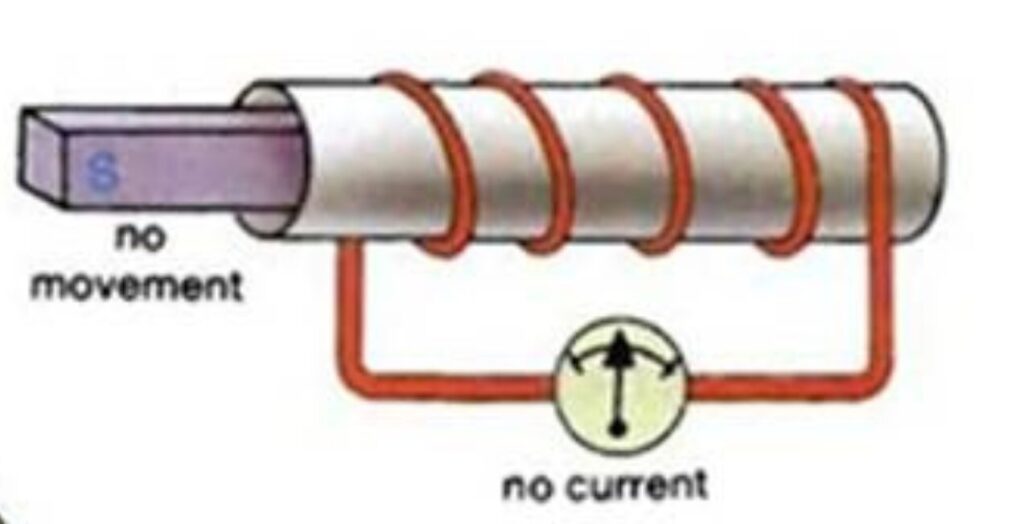
- Magnet inside the coil
- No Current flows
- Magnet outside the coil
- Current flows in opposite direction
Applications of Faraday’s Law of Induction
- Faraday’s laws are the foundation of modern electrical and electromagnetic systems. They are used in devices and technologies that generate, transfer, or use electricity efficiently.
Electric Generators:
Faraday’s law is the working principle of generators.
- When a coil rotates in a magnetic field, the changing magnetic flux induces current.
- Used in power stations to produce electricity on a large scale.
Transformers:
Used to transfer electrical energy from one circuit to another.
- A changing current in the primary coil induces voltage in the secondary coil.
- Helps step up or step down voltage for safe power transmission.
Wireless Chargers:
Used to charge phones and other gadgets without cables.
- A coil in the charger generates a changing magnetic field.
- This induces current in the device’s receiving coil.
Frequently Asked Questions
Solution:
Magnetic flux is the total magnetic field passing through a given area. It depends on the field strength, area size, and angle between them.
Solution:
- Increase the number of coil turns
- Use a stronger magnet
- Move the magnet or coil faster
- Use a soft iron core inside the coil
Solution:
Alternating current constantly changes direction, which creates a changing magnetic field, making it ideal for continuous induction in devices like transformers and motors.
Solution:
Soft iron enhances the magnetic field and helps direct it more efficiently through the coil, increasing the amount of induced EMF.
Solution:
Induction occurs when there is a change in magnetic flux—this can happen by moving a magnet, moving the coil, or changing the strength of the magnetic field.
Solution:
No. Only a changing magnetic field can induce current. A static field does not produce EMF.
Nuclear Energy: Fission and Fusion
Introduction
- Nuclear energy is generally the energy released from the nucleus (core) of an atom when it undergoes a nuclear reaction—either it can be fission or fusion.
Types of Nuclear Reactions:
1. Nuclear Fission:
- A heavy atom (like uranium-235 or plutonium-239) splits into two smaller atoms, releasing energy.
- Used in nuclear power plants.
2. Nuclear Fusion:
- Two light nuclei (like hydrogen) combine to form a heavier nucleus.
- Happens in sun and stars.
- Still under research for commercial use.
How is Nuclear Energy formed?
- Nuclear energy is formed from the nucleus (center) of an atom. There are two main ways this energy is created:
Nuclear Fission – Splitting Atoms
- A heavy atom (like uranium-235) is bombarded by a neutron.
- The atom splits into two smaller atoms.
- This split releases a huge amount of energy, along with more neutrons.
- These neutrons can hit other atoms, creating a chain reaction.
- Example:
Uranium-235 + neutron→Krypton + Barium + Energy + 3
Nuclear Fusion – Combining Atoms
- Two light atoms (like hydrogen isotopes) combine to form a heavier atom (like helium).
- This process releases energy because some mass is converted into energy (Einstein’s formula: E = mc²).
- ✅ Happens naturally in the sun and stars.
- ❌ Very hard to do on Earth – still under research.
Conclusion
Nuclear energy is formed when there is a change in the structure of atomic nuclei, either by fission or fusion.
- Fission is used in today’s nuclear power plants.
- Fusion is the energy of the future — cleaner, safer, and more powerful, but still in development.
Nuclear Fission
Definition:
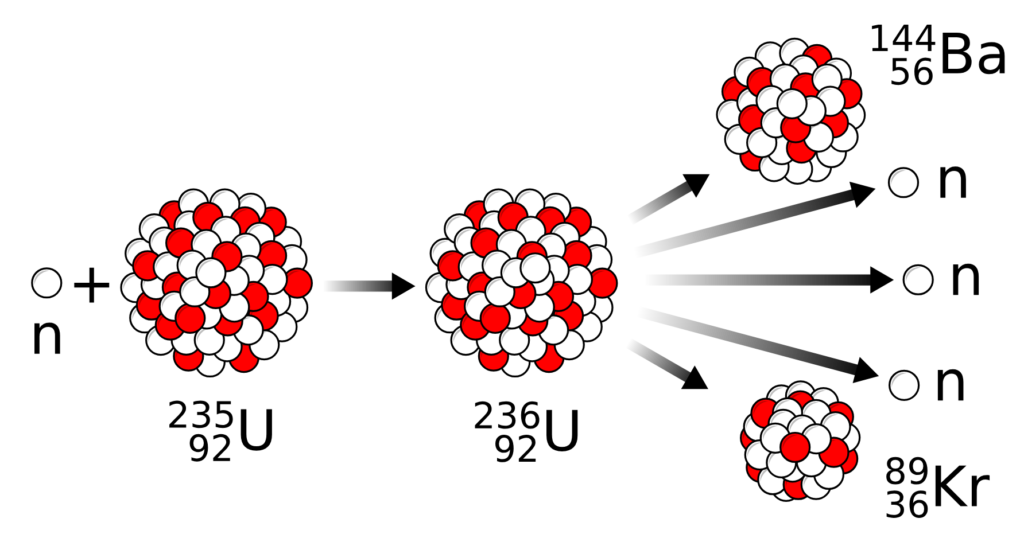
- Nuclear fission is a process in which a heavy atomic nucleus, such as uranium-235 or plutonium-239, is split into two lighter nuclei, releasing a large amount of energy, along with free neutrons and gamma radiation.
How it works:
- Let say a neutron hits a uranium-235 atom.
- The uranium atom becomes unstable and splits.
- This produces:
- Two smaller nuclei (e.g., krypton and barium),
- 3 more neutrons, and
- A large amount of heat energy.
🔄 Chain Reaction:
- The neutrons released can hit more
U235+ n → Ba144 + Kr89 + 3n + Energy
Where the energy is used:
- Nuclear power plants (for generating electricity).
- Nuclear submarines and ships.
- Atomic bombs (uncontrolled chain reaction).
Practical Applications of Nuclear Fission
- Nuclear fission has several real-world applications, especially because it produces a large amount of energy from a small amount of fuel. Here’s a detailed list:
Electricity Generation (Nuclear Power Plants)
- The most common application.
- Nuclear reactors use fission of Uranium-235 to produce heat.
- This heat is used to turn water into steam.
- Steam rotates turbines, which drive generators to produce electricity.
📍 Example:
- Bruce Nuclear Generating Station in Canada

Nuclear-Powered Submarines and Ships
- Fission reactors are used in naval vessels for propulsion.
- They can stay underwater or travel long distances without refueling for years.
📍 Example:
- USS Enterprise (USA) – first nuclear-powered aircraft carrier

Scientific Research
- Research reactors use controlled fission to study neutron behavior, material properties, and nuclear physics.
- Helps in material testing and developing better fuels.

Nuclear Fusion
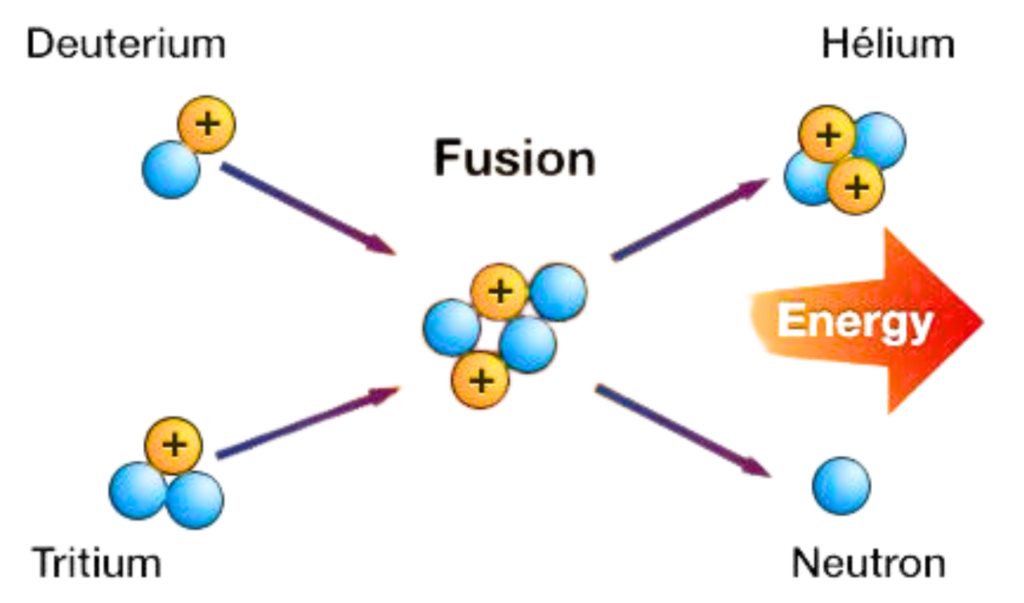
Definition:
- Nuclear fusion is a reaction where two light atomic nuclei (like hydrogen isotopes) fuse together to form a heavier nucleus, releasing massive amounts of energy.
How it works:
- At extremely high temperatures and pressures, two hydrogen nuclei (deuterium and tritium) overcome their repulsion and combine.
- This forms helium and releases energy.
- Some mass is converted into energy, based on Einstein’s formula:
E = mc2
where,
- E = energy,
- m = mass lost,
- c = speed of light.
Where it Happens:
- In the core of stars, including the Sun.
- Under research in labs on Earth (e.g., ITER project) for clean energy.
- 📌 Equation Example:
D + T → He +n + Energy
Practical Applications of Nuclear Fusion
- Nuclear fusion is the process where two light atomic nuclei combine to form a heavier nucleus, releasing enormous energy. Though it’s not yet commercially used on Earth, fusion powers the Sun and stars and is under active research for clean energy.
The Sun and Other Stars
- The most natural and perfect example of nuclear fusion.
Example:
- In the Sun, hydrogen nuclei (protons) fuse to form helium and release energy.
- This energy reaches Earth as sunlight and heat.
- Fusion keeps stars shining for billions of years.
4H → He + Energy
Hydrogen Bomb (Fusion Bomb)
- Fusion is used in thermonuclear weapons, where it releases even more energy than fission bombs.
Example:
- Tsar Bomba (1961) – Largest hydrogen bomb ever tested by Russia.
- Works by fusing deuterium and tritium after triggering with a fission bomb.

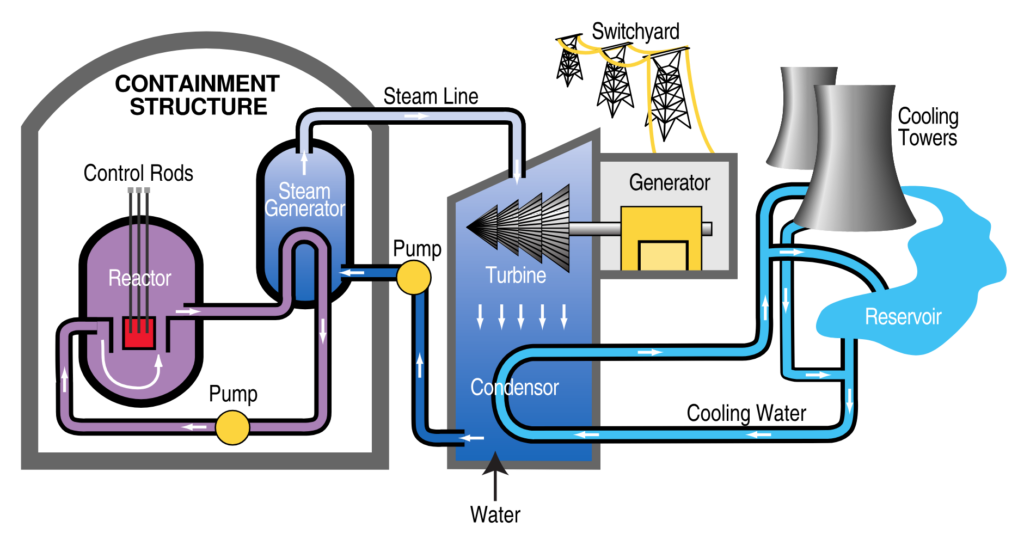
Nuclear Power Plant
- A Nuclear Power Plant is a facility that generates electricity through controlled nuclear fission reactions, typically using uranium or plutonium as fuel.
- These plants produce large amounts of energy with relatively low greenhouse gas emissions, making them a key part of the clean energy debate.
How it works:
Nuclear Fission:
- Uranium-235 (or plutonium-239) atoms are split in a reactor core, releasing heat.
- This process is sustained by a chain reaction, controlled using neutron-absorbing materials (like control rods).
Heat Generation:
- The fission reaction heats water in the primary coolant loop, which remains under high pressure to prevent boiling.
Steam Production (via Heat Exchanger):
- The hot coolant transfers heat to a secondary water loop, turning it into steam (in a steam generator).
Electricity Generation:
- The steam drives a turbine, which spins a generator to produce electricity.
- After passing through the turbine, the steam is cooled back into water in a condenser (often using a cooling tower or nearby water source).
Waste & Safety Systems:
- Spent fuel (radioactive waste) is stored on-site in cooling pools or dry casks.
- Multiple safety systems (containment structures, emergency cooling) prevent radiation leaks.
Types of Nuclear Reactors:
- Pressurized Water Reactor (PWR) – Most common; uses high-pressure water as coolant/moderator.
- Boiling Water Reactor (BWR) – Simpler design; steam is produced directly in the reactor.
- Advanced Reactors –
- Fast Breeder Reactors (FBRs) – Produce more fissile material than they consume.
- Small Modular Reactors (SMRs) – Compact, scalable designs for flexible deployment.
- Molten Salt Reactors (MSRs) – Use liquid fuel for potentially safer operation.
Difference between Nuclear Fission and Fusion

Frequently Asked Questions
Solution:
- Two smaller nuclei (called fission fragments)
- 3 neutrons (on average)
- Energy (in the form of heat and radiation)
Solution:
Yes, in hydrogen bombs and experimental reactors (like ITER), but not yet in a controlled, power-generating way.
Solution:
- Uranium-235
- Plutonium-239
Solution:
- Deuterium (from water)
- Tritium (produced from lithium)
Solution:
Because fusion needs extremely high temperatures and pressures (like those inside stars), it is very hard to control.
Researchers are working on it (e.g., ITER project), but it’s not ready for electricity production yet.
Solution:
- Fission-based nuclear energy is not renewable (limited uranium)
- Fusion-based energy could be considered nearly unlimited and clean, but it’s not yet available for use


