Types of Substance – GCSE Chemistry
Introduction
- A Substance is a form of matter that has a constant composition and distinct properties. Substances can exist as pure elements, compounds, or mixtures, and they are classified based on their chemical structure and bonding.
Understanding substances helps in:
- Chemical reactions (how substances interact).
- Material science (developing new materials).
- Medicine & Pharmaceuticals (drug formulation).
Formation of Different Substances
Method Used:
- The Dot-and-Cross (Lewis) diagram is a simple way to represent how atoms bond to form substances by showing the valence electrons involved in bonding. Below, we illustrate the formation of ionic and covalent (molecular) compounds using this method.
Example related to Ionic Substances:
- Formed when a metal transfers electrons to a nonmetal, creating oppositely charged ions that attract each other.
Example: Formation of Sodium Chloride (NaCl)
- Sodium (Na) – Metal (Group 1) with 1 valence electron.
- Chlorine (Cl) – Nonmetal (Group 17) with 7 valence electrons.
Dot-and-Cross Diagram:
1. Sodium loses 1 electron → forms Na⁺.
2. Chlorine gains 1 electron → forms Cl⁻.
3. Ions attract electrostatically to form NaCl.
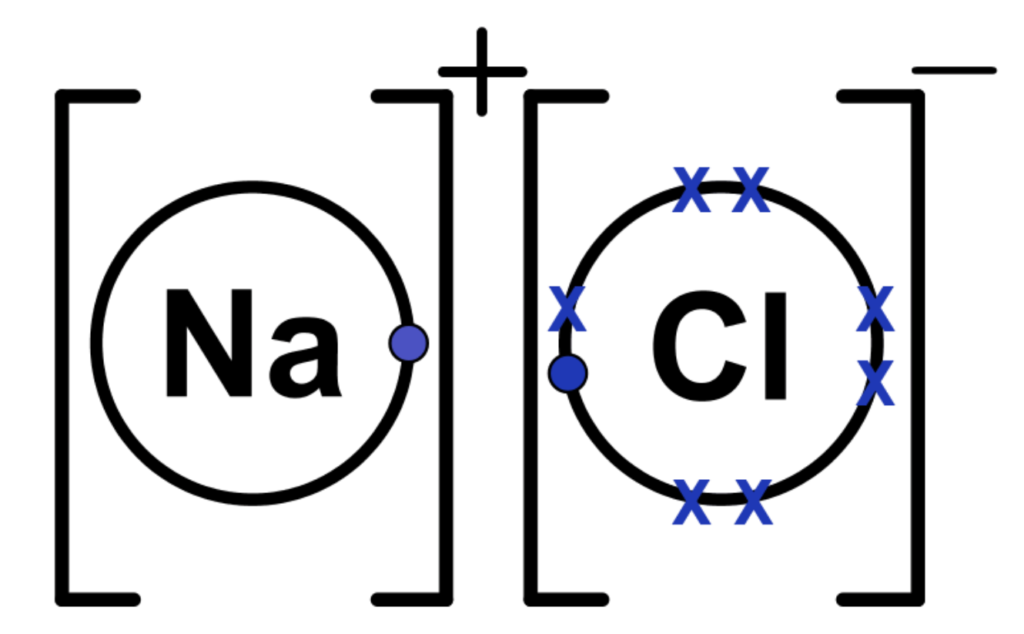
Key Points:
- Dot (•) = Sodium’s electron.
- Cross (×) = Chlorine’s electron.
- Brackets [ ] = show the ion’s charge.
Example related to Covalent Substances:
- Formed when nonmetals share electrons to achieve a stable electron configuration.
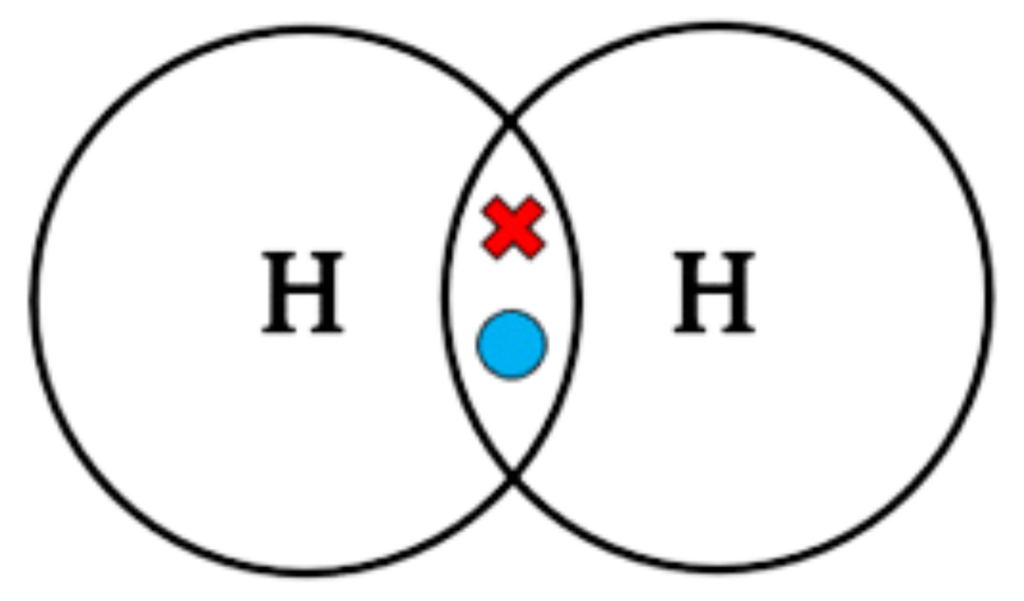
Example: Formation of Hydrogen (H₂)
- Each hydrogen atom has 1 valence electron.
- They share 1 pair of electrons to form a single covalent bond.
Dot-and-Cross Diagram:
Key Points:
✔ Single covalent bond (1 shared pair of electrons).
✔ No lone pairs (all electrons are shared).
✔ Linear shape (only two atoms).
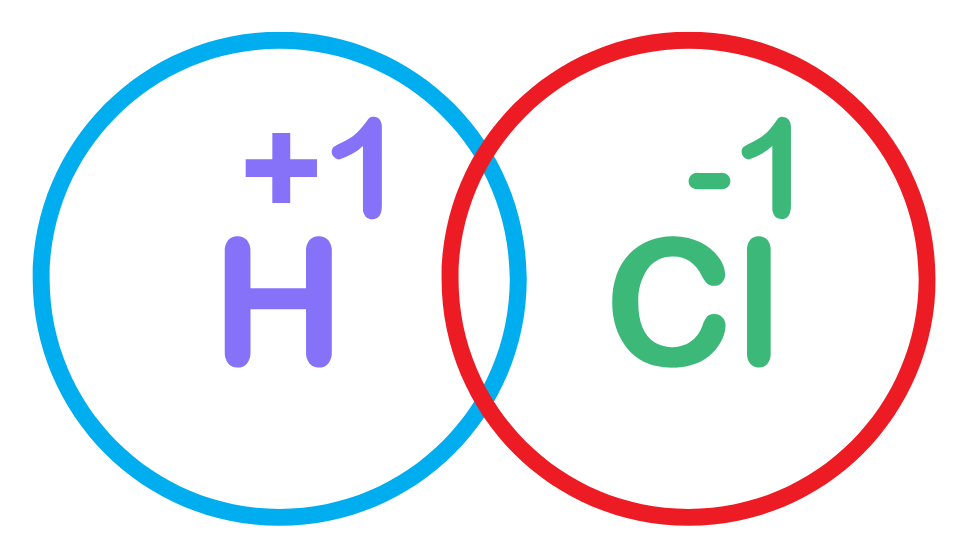
More Examples related to Dot and Cross Method
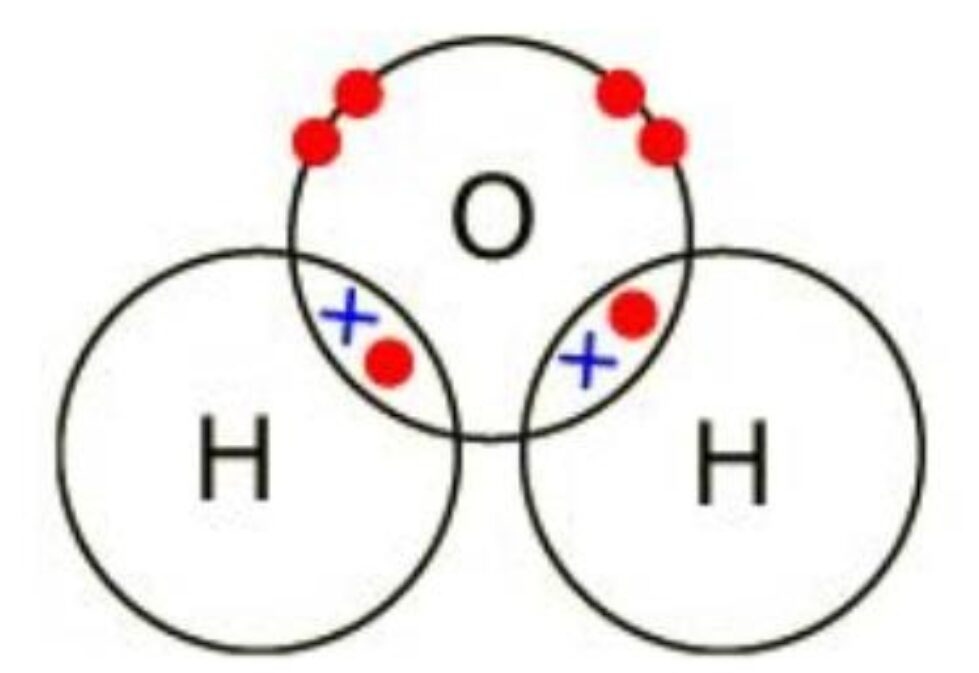
Example 1: Dot and Cross diagram of H2O
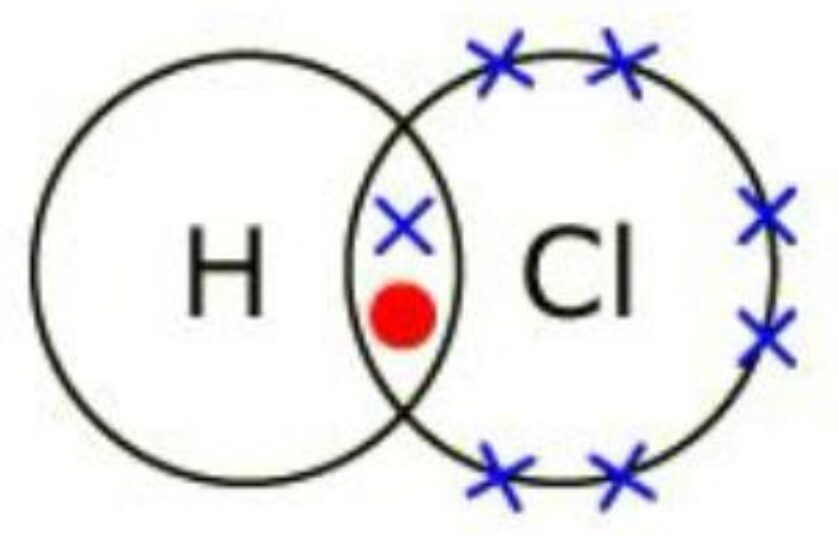
Example 2: Dot and Cross diagram of HCl
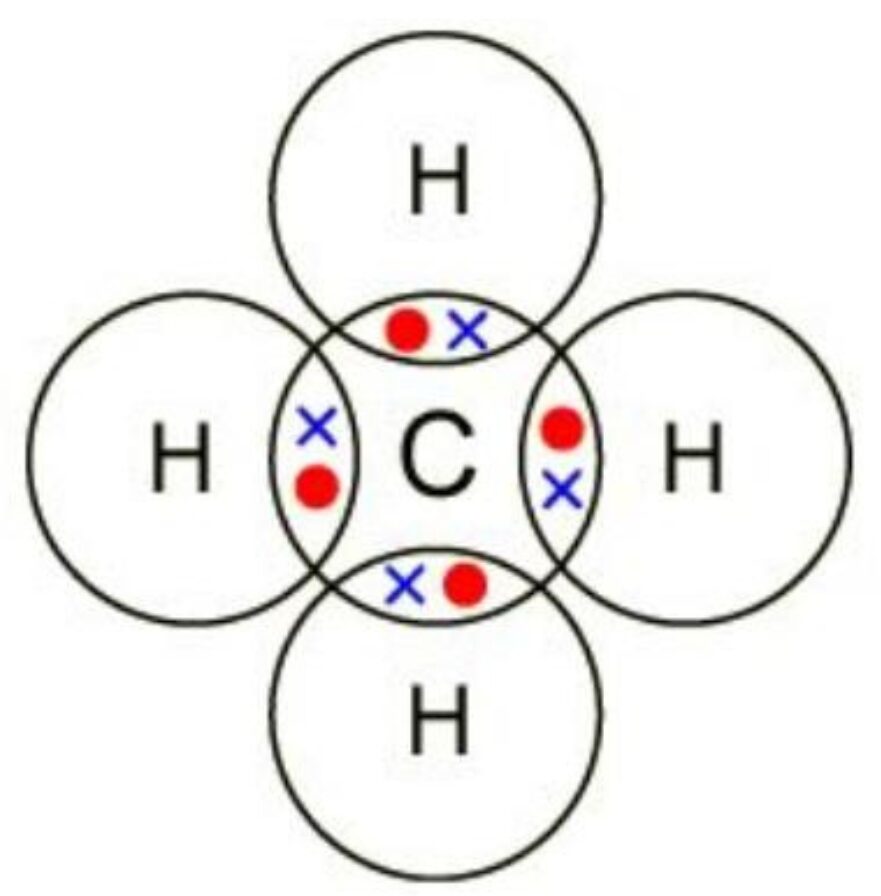
Example 3: Dot and Cross diagram of CH4
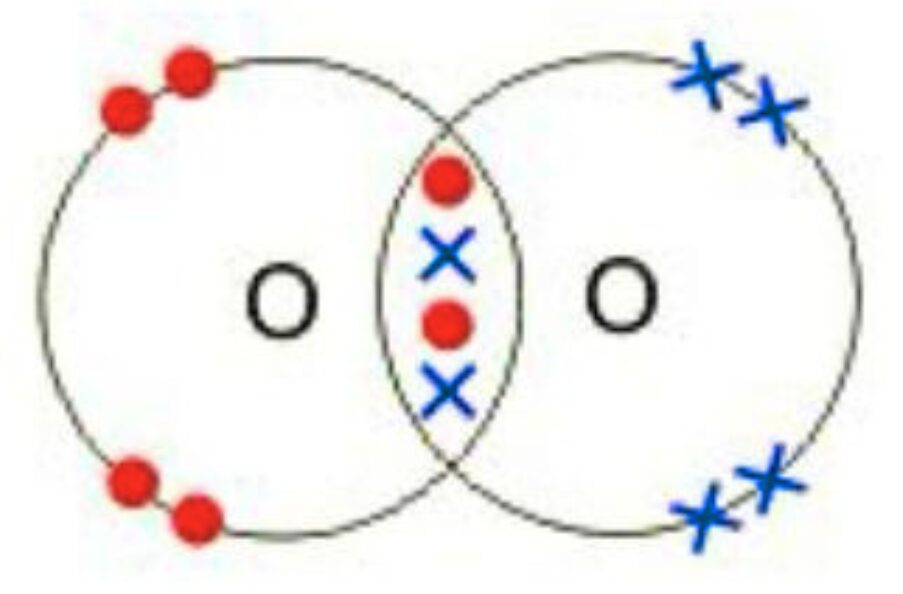
Example 4: Dot and Cross diagram of O2
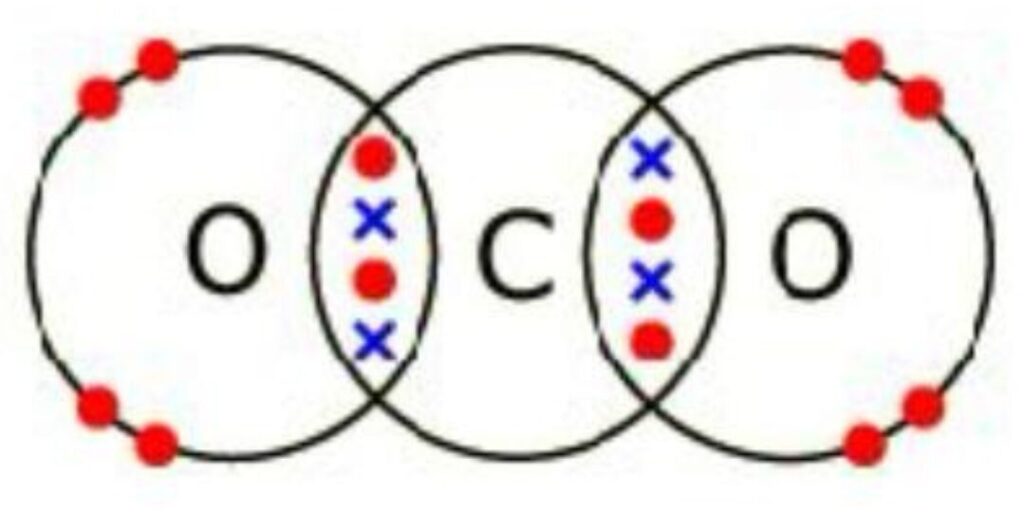
Example 5: Dot and Cross diagram of CO2
Properties of Ionic and Simple Molecular Compounds
Properties of Ionic Compounds:
Structure:
- Made of positive and negative ions arranged in a giant lattice.
- Formed by electron transfer from metals to nonmetals.
Physical Properties:
- High melting and boiling points (strong electrostatic forces between ions).
- Solid at room temperature (except some molten salts).
- Brittle (layers shift under stress, causing repulsion).
- Soluble in water (polar solvents break ionic bonds).
- Conduct electricity when molten or dissolved (free-moving ions).

Chemical Properties:
- Form crystals (e.g., NaCl, MgO).
- Undergo electrolysis when molten or in solution.
Properties of Simple Molecular Compounds:
Structure:
- Made of neutral molecules with shared electrons.
- Formed by electron sharing between nonmetals.

Physical Properties:
- Low melting and boiling points (weak intermolecular forces).
- Gases, liquids, or soft solids at room temperature (e.g., O₂, H₂O, wax).
- Poor electrical conductivity (no free ions or electrons).
- Most are insoluble in water (except polar molecules like sugar).
Chemical Properties:
- Exist as discrete molecules (e.g., CO₂, CH₄).
- Volatile (easily evaporate due to weak forces).
Giant Covalent Structures
- Giant Covalent structures are three-dimensional networks of atoms bonded together by strong covalent bonds in a repeating pattern. Unlike simple molecules, these structures extend indefinitely, forming rigid solids with unique properties.
Key Features:
✔ No individual molecules – The entire structure acts as one giant molecule.
✔ Very strong bonds – High energy required to break covalent bonds.
✔ High melting/boiling points – Due to extensive covalent bonding.
✔ Insoluble in water – Nonpolar and too tightly bonded to dissolve.
✔ Variable conductivity – Most are insulators, except graphite (conducts electricity).
Examples of Giant Covalent Structures:
- Diamond
- Graphite
- Silicon
- Carbide
- Quartz
Let us understand some of the examples of Giant Covalent Structures i.e,
- Diamond
- Graphite
Diamond
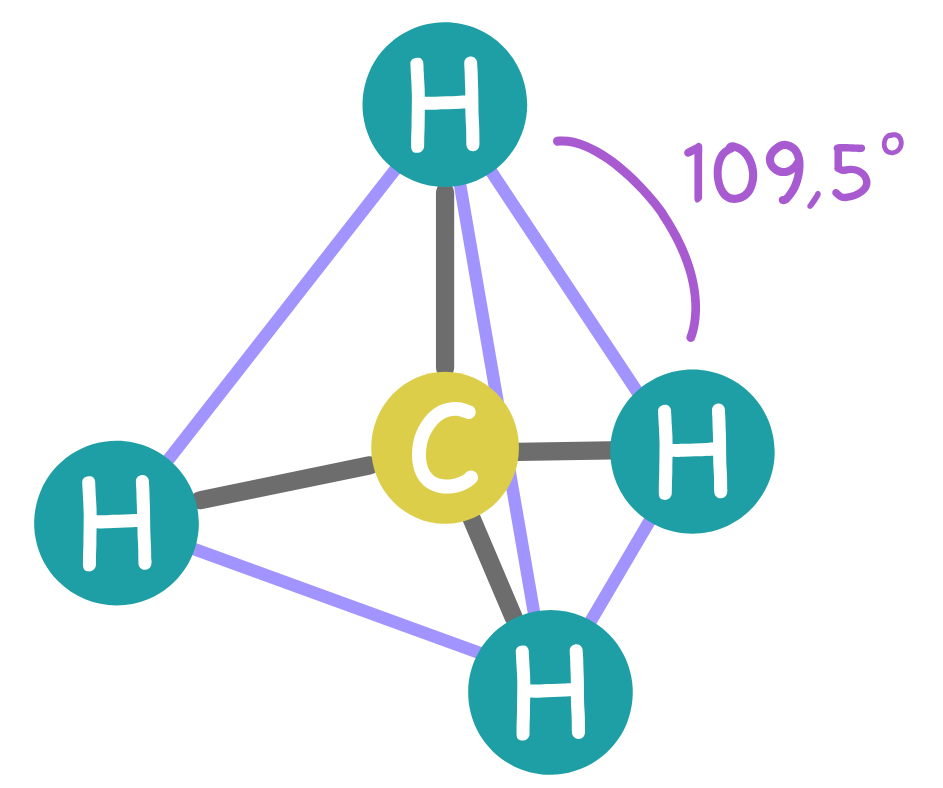
- Diamond is a giant covalent structure composed entirely of carbon atoms, arranged in a rigid 3D lattice. Each carbon forms four strong covalent bonds in a tetrahedral geometry, creating an exceptionally hard and thermally conductive material.
1. Tetrahedral Bonding
- Each carbon atom is covalently bonded to 4 other carbons
- Bond angle: 109.5° (perfect tetrahedral symmetry)
- Bond length: 0.154 nm (very short, contributing to hardness)
2. 3D Network Structure
- Infinite repetition of tetrahedral units
- No weak points – bonds are uniformly strong in all directions
- No free electrons – all valence electrons are used in σ-bonds
3. Unit Cell (Cubic Crystal System)
- Face-centered cubic (FCC) lattice with additional atoms
- 8 atoms per unit cell (4 from FCC + 4 internal)
- Coordination number: 4 (each C has 4 nearest neighbors)
Graphite
- Graphite is a layered giant covalent structure composed entirely of carbon atoms, arranged in stacked hexagonal sheets. Unlike diamond, each carbon forms three covalent bonds in a trigonal planar geometry, leaving delocalized electrons that enable unique properties like electrical conductivity.
1. Hexagonal Layered Structure
- Each carbon atom is covalently bonded to 3 others in a 2D plane
- Bond angle: 120° (perfect hexagonal symmetry)
- Bond length: 0.142 nm (shorter than diamond due to partial double-bond character)
2. Interlayer Weak Forces
- Layers held by van der Waals forces (spacing: 0.335 nm)
- Easy layer sliding → gives graphite its lubricating properties
- Anisotropic behavior (properties differ along vs. across layers)
3. Delocalized Electrons
- One free electron per carbon forms a π-electron cloud
- Enables electrical conductivity within planes
- Absorbs visible light → opaque black appearance.

Important Note:
- Graphite acts a a lubricant due to these reasons:
- Layered structure with strong covalent bonds within sheets but weak van der Waals forces between sheets, allowing easy sliding.
- Shearing force makes layers slip past each other, reducing friction.
- Stable at high temps (unlike oils) and works in dry conditions.
- Used in locks, engines, and pencils.
Allotropes of Carbon such as Fullerenes and Graphene
- Carbon exists in different structural forms called allotropes, each with unique properties due to variations in atomic arrangement and bonding.
Let us discuss in brief about the two allotropes i.e,
- Fullerene and Graphene:
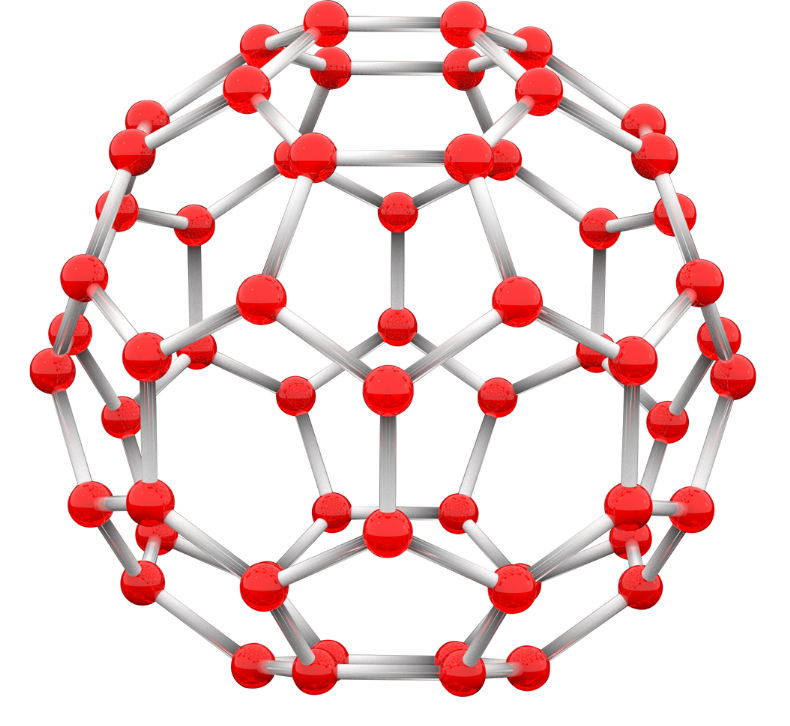
Fullerene
- Closed 3D structures (sp² hybridized carbon)
- Examples:
- C₆₀ (soccer ball shape)
- Carbon nanotubes (cylindrical tubes)
- Curvature introduces strain in bonds
- Produced by vaporizing carbon (laser/arc methods)
Graphene
- Single layer of graphite (flat 2D sheet)
- Perfect hexagonal lattice (no pentagons)
- No curvature → ideal sp² bonding
- Exfoliated from graphite (Scotch tape method)

NOTE: Graphene is essentially an “unrolled” carbon nanotube or a “single layer” of graphite, while fullerenes are its “rolled/closed” cousins!
Frequently Asked Questions
Most lack free ions/electrons. Exceptions: Graphite (delocalized electrons in layers). Polar covalent compounds (e.g., HCl in water) dissociate into ions.
No! Giant covalent (e.g., diamond) are extremely strong. Weakness applies to intermolecular forces in simple molecules (e.g., CO₂).
- Ethene (C₂H₄): A simple hydrocarbon gas (alkene) with a double bond between two carbon atoms.
- Polyethene (Polyethylene, (C₂H₄)ₙ): A polymer formed by linking thousands of ethene monomers into long chains.
Metallic bonding: Positive metal ions in a “sea of delocalized electrons” allow layers to slide past each other without shattering.
- Heating: Increases malleability (reduces hardness, eases dislocation movement).
- Cooling: Decreases malleability (makes metals brittle; e.g., frozen steel).
- Diamond: Large band gap (5.5 eV) → no light absorption in visible spectrum.
- Graphite: Delocalized electrons absorb all light wavelengths.

